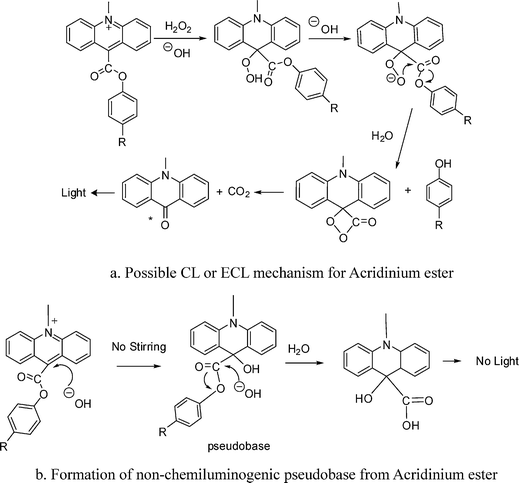
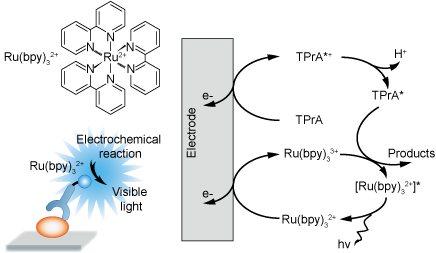
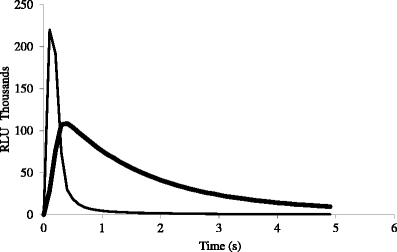
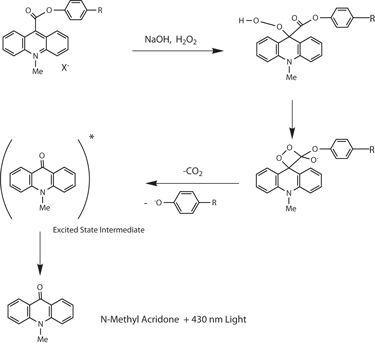
Strongly Chemiluminescent Acridinium Esters under Neutral Conditions: Synthesis, Properties, Determination, and Theoretical Study - ScienceDirect
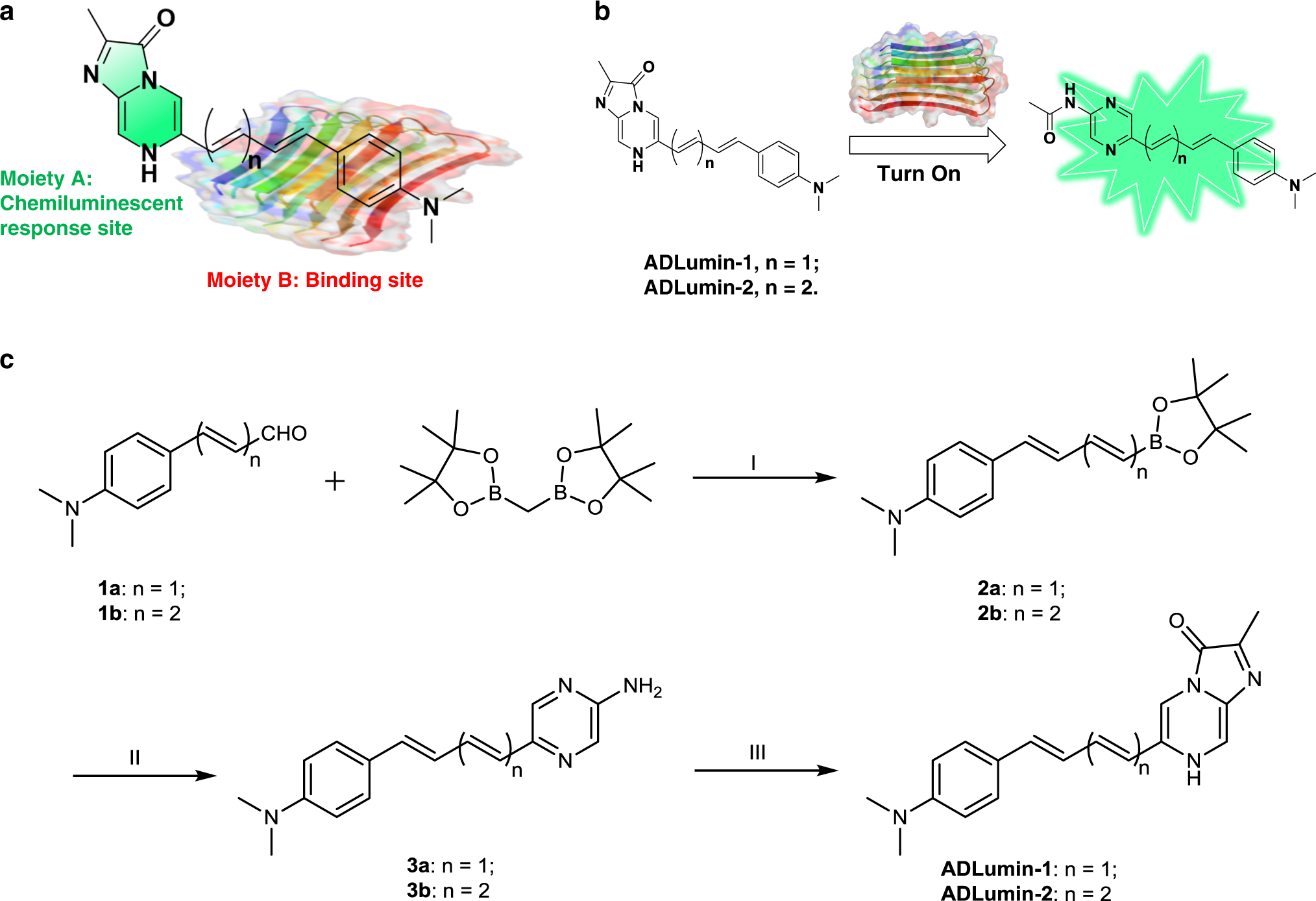
Turn-on chemiluminescence probes and dual-amplification of signal for detection of amyloid beta species in vivo | Nature Communications

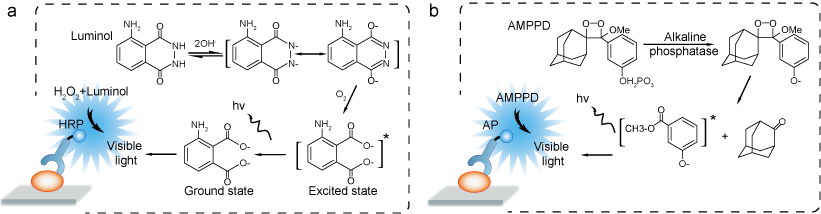
Scheme 2 (a) Mechanism of oxidative chemiluminescence from luminol via... | Download Scientific Diagram
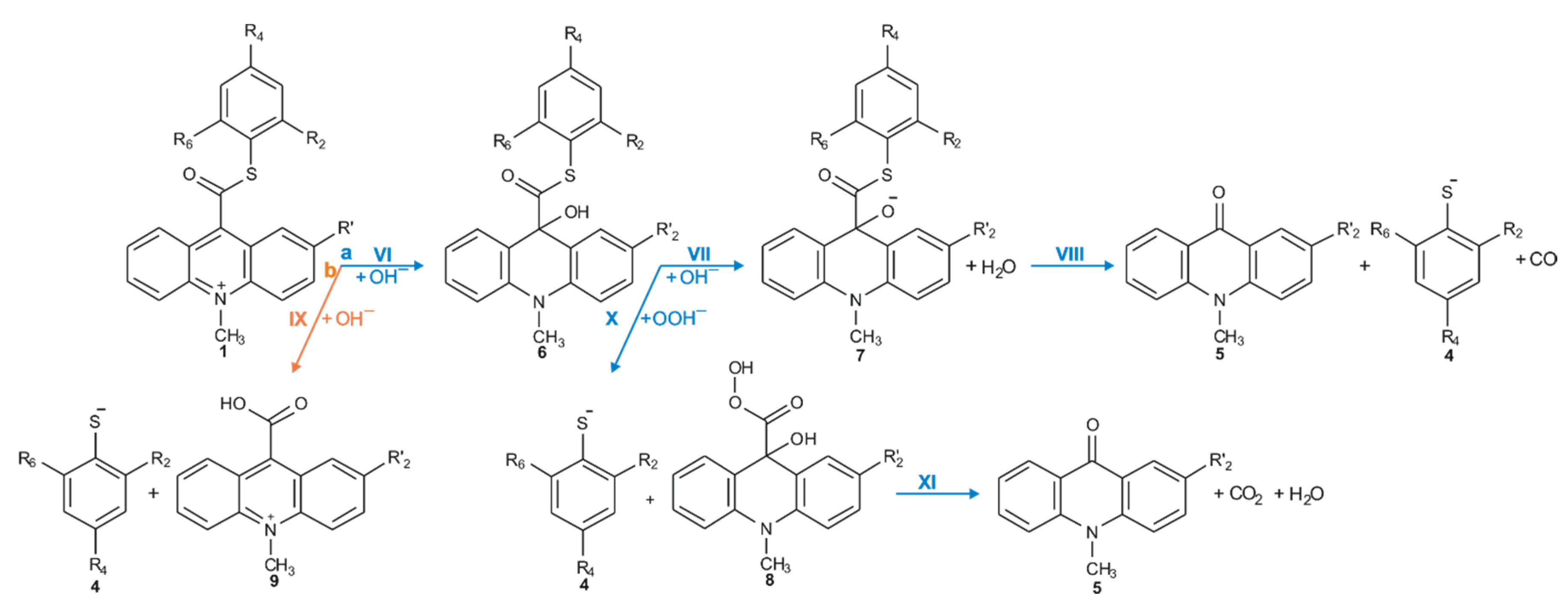
IJMS | Free Full-Text | Computational Insights on the Mechanism of the Chemiluminescence Reaction of New Group of Chemiluminogens—10-Methyl-9-thiophenoxycarbonylacridinium Cations | HTML
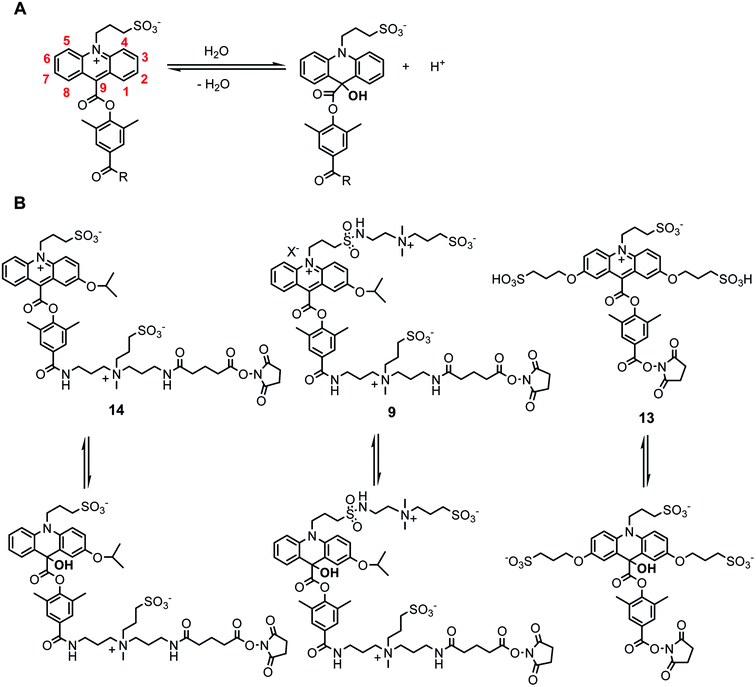
Synthesis and properties of differently charged chemiluminescent acridinium ester labels - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB27190G

Synthesis and properties of chemiluminescent acridinium ester labels with fluorous tags - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB00456F
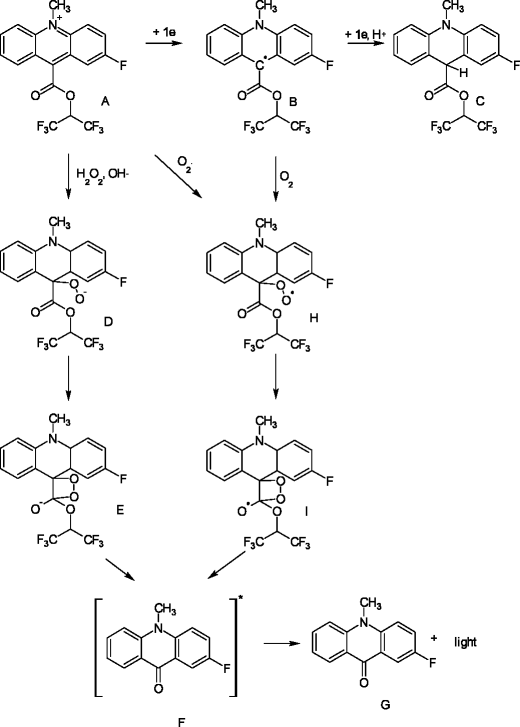
Chemiluminescent reductive acridinium triggering (CRAT)--mechanism and applications - Document - Gale Academic OneFile

IJMS | Free Full-Text | Computational Insights on the Mechanism of the Chemiluminescence Reaction of New Group of Chemiluminogens—10-Methyl-9-thiophenoxycarbonylacridinium Cations | HTML

Strongly Chemiluminescent Acridinium Esters under Neutral Conditions: Synthesis, Properties, Determination, and Theoretical Study | The Journal of Organic Chemistry
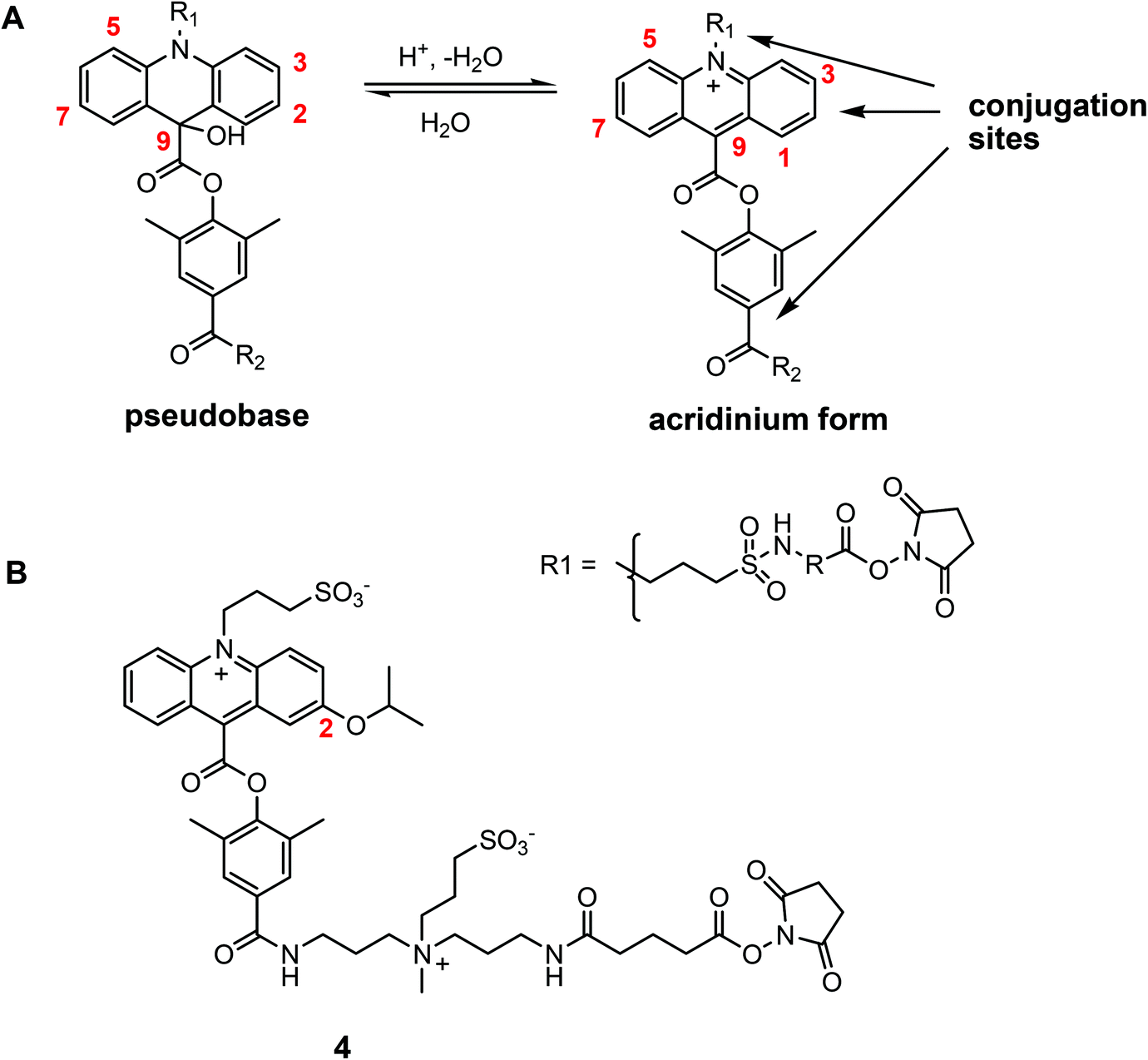
A comparison of chemiluminescent acridinium dimethylphenyl ester labels with different conjugation sites - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB02528H

US6673560B1 - Measurement of hydride using chemiluminescent acridinium compounds and applications thereof - Google Patents