![Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences](https://royalsocietypublishing.org/cms/asset/0ea92727-dfe5-4dad-b4fd-c68f2d8d3d5e/rspa20190238f03.jpg)
Facile synthesis of 2-arylimidazo[1,2-a]pyridines catalysed by DBU in aqueous ethanol | Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences
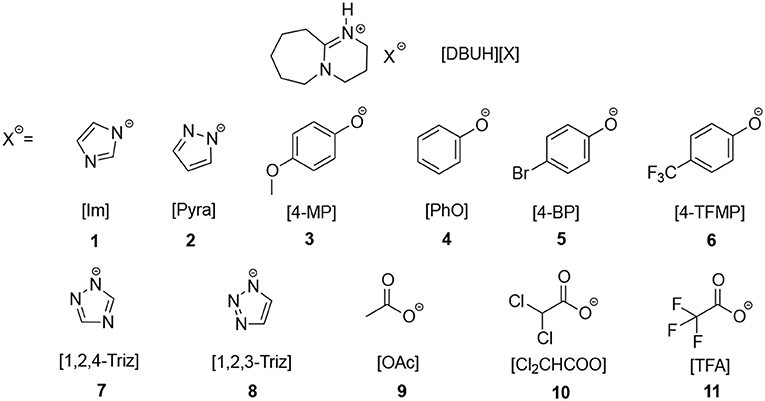
Frontiers | CO2 Absorption by DBU-Based Protic Ionic Liquids: Basicity of Anion Dictates the Absorption Capacity and Mechanism | Chemistry

Unprecedented cooperative DBU-CuCl2 catalysis for the incorporation of carbon dioxide into homopropargylic amines leading to 6-methylene-1,3-oxazin-2-ones - ScienceDirect
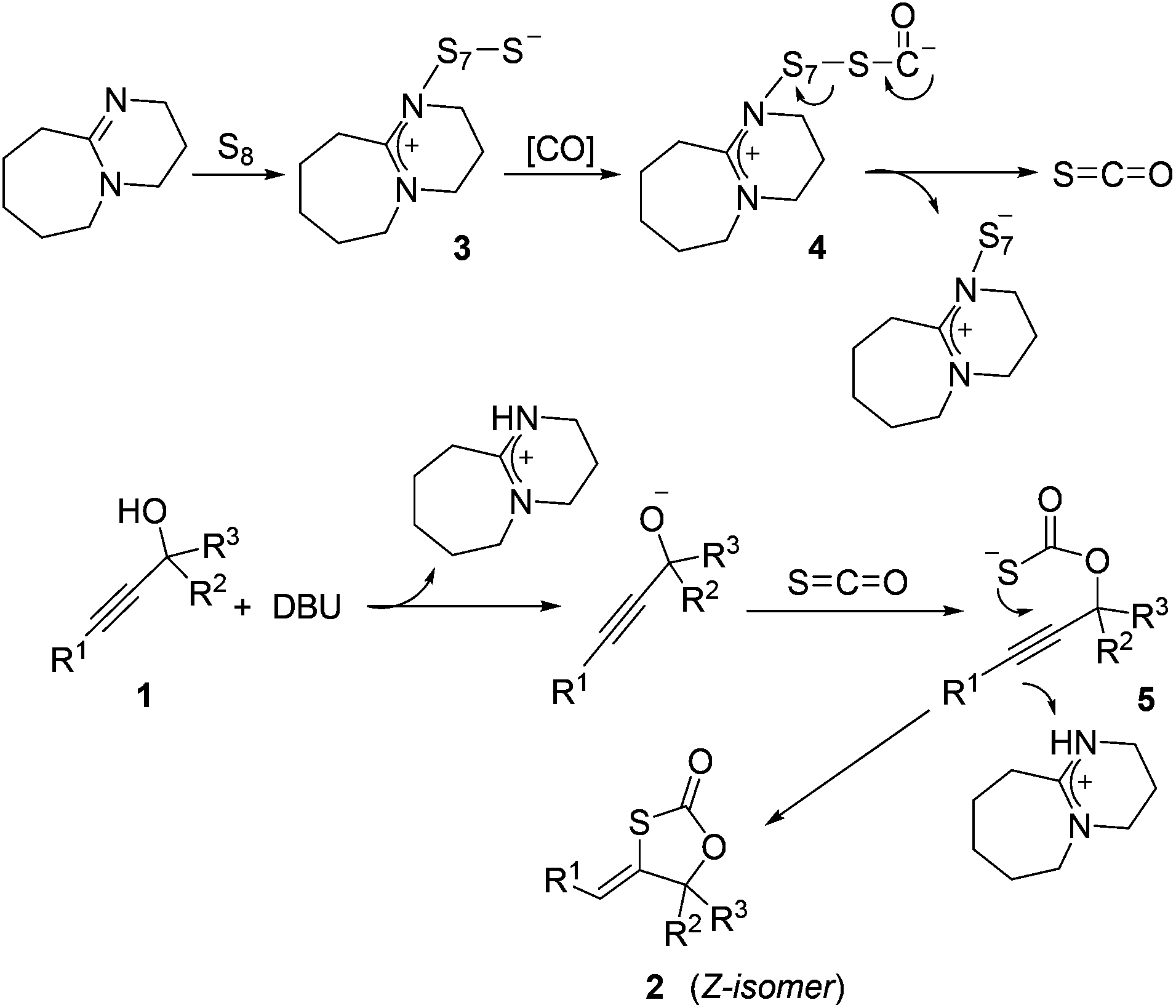
DBU-promoted carbonylative synthesis of 1,3-oxathiolan-2-ones from propargylic alcohols with TFBen as the CO source - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB03145A

Umpolung cyclization reaction of N -cinnamoylthioureas in the presence of DBU - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB02066C
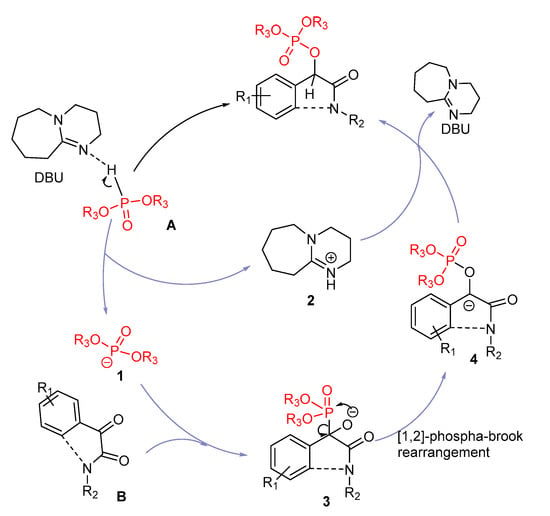
Catalysts | Free Full-Text | DBU Catalyzed Phospho-Aldol-Brook Rearrangement for Rapid Preparation of α-Phosphates Amide in Solvent-Free Conditions | HTML
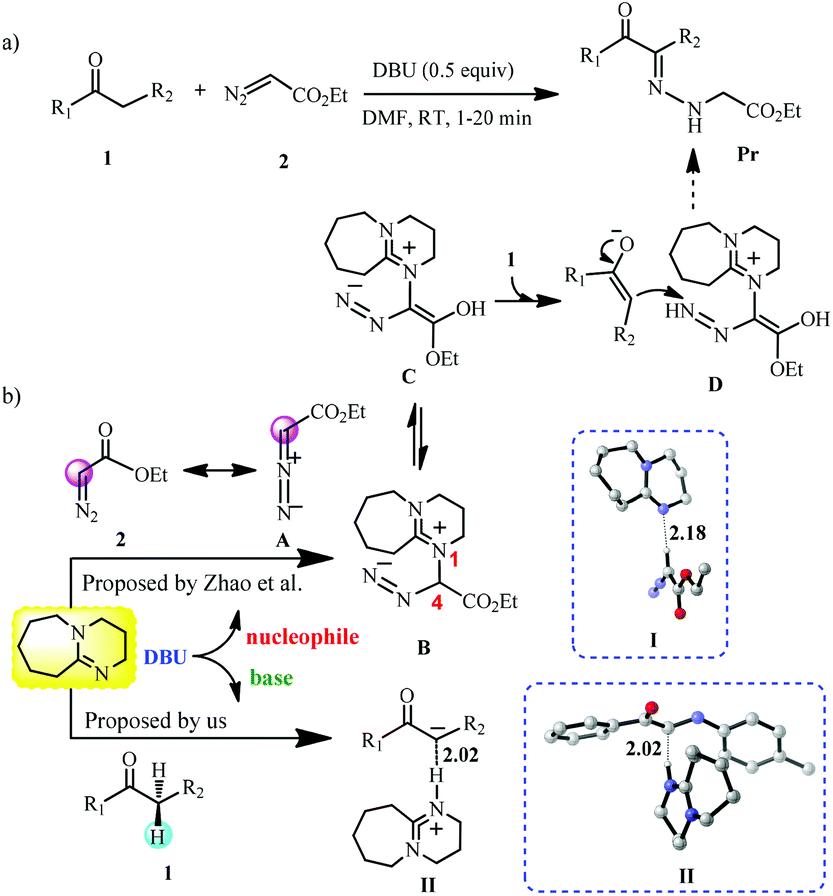
Mechanistic investigation-inspired activation mode of DBU and the function of the α-diazo group in the reaction of the α-amino ketone compound and EDA ... - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO00602H

Unprecedented, selective Nef reaction of secondary nitroalkanes promoted by DBU under basic homogeneous conditions
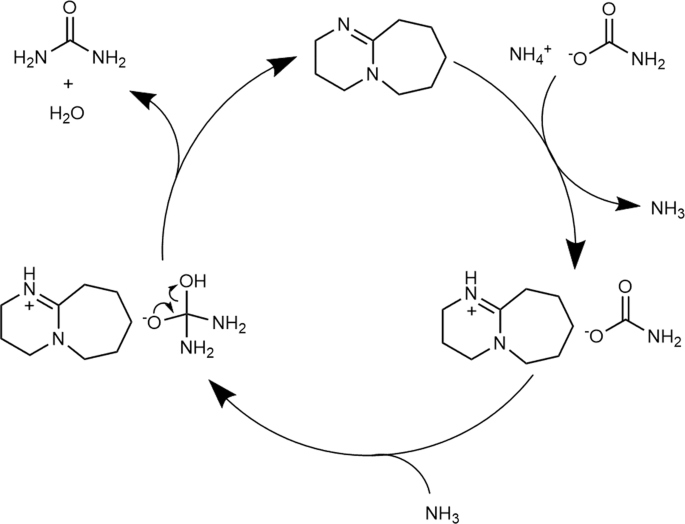
Organic bases catalyze the synthesis of urea from ammonium salts derived from recovered environmental ammonia | Scientific Reports
![Scheme 7. Proposed mechanism for the 1,8-diazabicyclo[5.4.0]undec-7-ene... | Download Scientific Diagram Scheme 7. Proposed mechanism for the 1,8-diazabicyclo[5.4.0]undec-7-ene... | Download Scientific Diagram](https://www.researchgate.net/publication/338016321/figure/fig27/AS:846575794393088@1578850806233/Scheme-7-Proposed-mechanism-for-the-1-8-diazabicyclo540undec-7-ene-DBU-mediated.png)
Scheme 7. Proposed mechanism for the 1,8-diazabicyclo[5.4.0]undec-7-ene... | Download Scientific Diagram
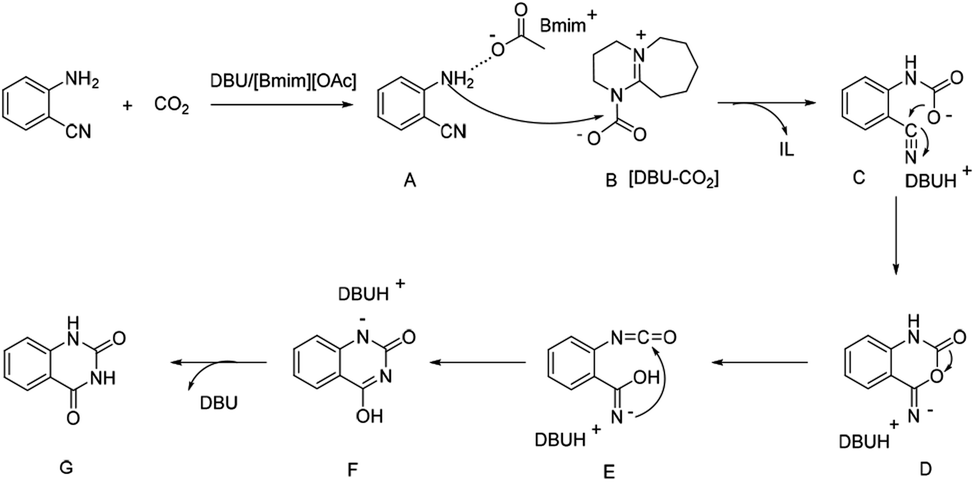
DBU coupled ionic liquid-catalyzed efficient synthesis of quinazolinones from CO 2 and 2-aminobenzonitriles under mild conditions - RSC Advances (RSC Publishing) DOI:10.1039/D0RA00194E

Pd-Catalyzed C-N Coupling Reactions Facilitated by Organic Bases: Mechanistic Investigation Leads to Enhanced Reactivity in the Arylation of Weakly Binding Amines. - Abstract - Europe PMC

SciELO - Brasil - Mechanistic Investigation of DBU-Based Ionic Liquids for Aza-Michael Reaction: Mass Spectrometry and DFT Studies of Catalyst Role Mechanistic Investigation of DBU-Based Ionic Liquids for Aza-Michael Reaction: Mass Spectrometry
Proposed mechanisms for the DBU catalyzed urea-synthesis reaction from... | Download Scientific Diagram
![PDF] 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU)-promoted decomposition of difluorocarbene and the subsequent trifluoromethylation. | Semantic Scholar PDF] 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU)-promoted decomposition of difluorocarbene and the subsequent trifluoromethylation. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/05a5a185fd081b7b27d083bf113c654cb755fa96/3-Figure6-1.png)
PDF] 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU)-promoted decomposition of difluorocarbene and the subsequent trifluoromethylation. | Semantic Scholar

![The grafting copolymerization of PLA onto xylan in [Amim]Cl with... | Download Scientific Diagram The grafting copolymerization of PLA onto xylan in [Amim]Cl with... | Download Scientific Diagram](https://www.researchgate.net/publication/315914896/figure/fig4/AS:668320676200457@1536351474691/The-grafting-copolymerization-of-PLA-onto-xylan-in-AmimCl-with-organic-catalyst-DBU-A_Q640.jpg)

![Synthesis of Azobenzenes Using N-Chlorosuccinimide and 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU) Synthesis of Azobenzenes Using N-Chlorosuccinimide and 1,8-Diazabicyclo[5.4.0]undec-7-ene (DBU)](https://www.organic-chemistry.org/abstracts/lit6/017o.gif)