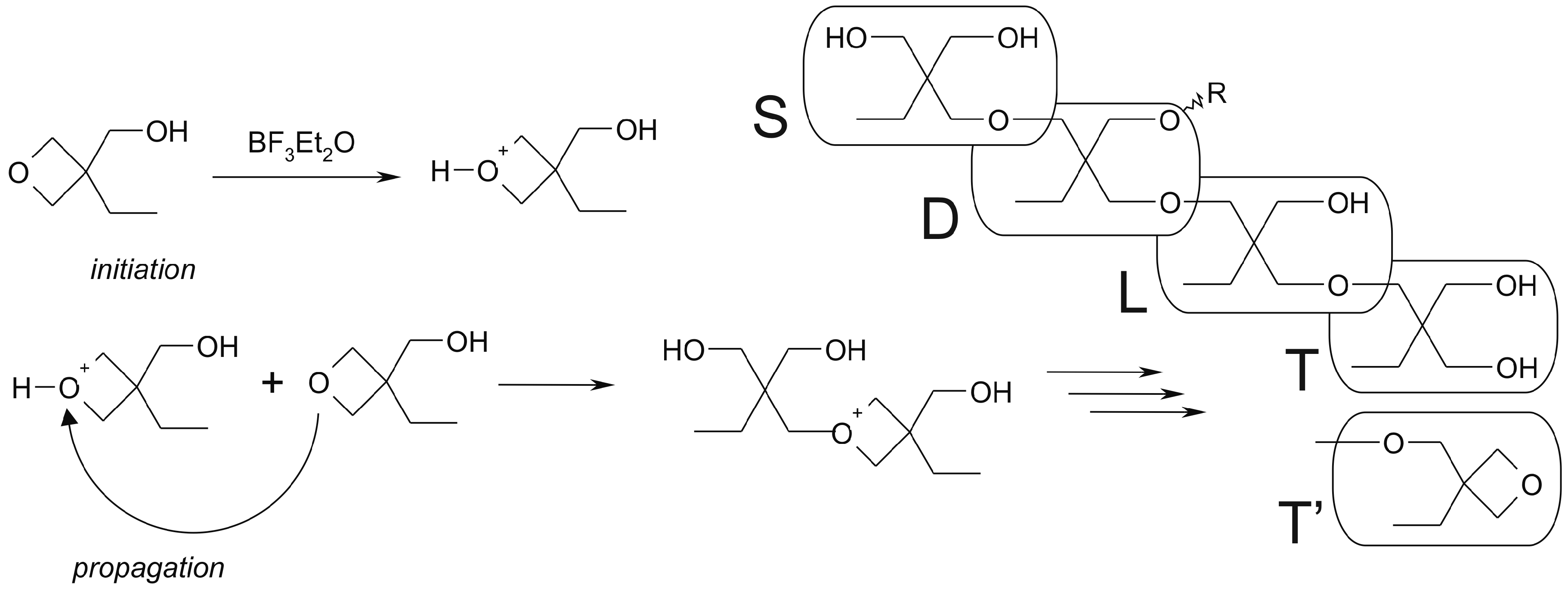
![PDF] A comparative study of amide-bond forming reagents in aqueous media – Substrate scope and reagent compatibility | Semantic Scholar PDF] A comparative study of amide-bond forming reagents in aqueous media – Substrate scope and reagent compatibility | Semantic Scholar](https://ai2-s2-public.s3.amazonaws.com/figures/2017-08-08/5f8253ff990797847606bac48011d4a849802994/4-Figure2-1.png)
PDF] A comparative study of amide-bond forming reagents in aqueous media – Substrate scope and reagent compatibility | Semantic Scholar

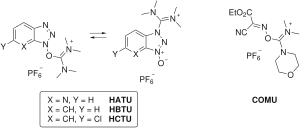
Scheme 1 Fmoc-depsidipeptide synthesis, whereby (a) Fmoc-peptide in... | Download Scientific Diagram

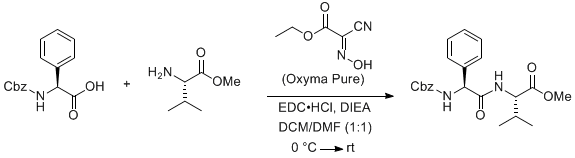
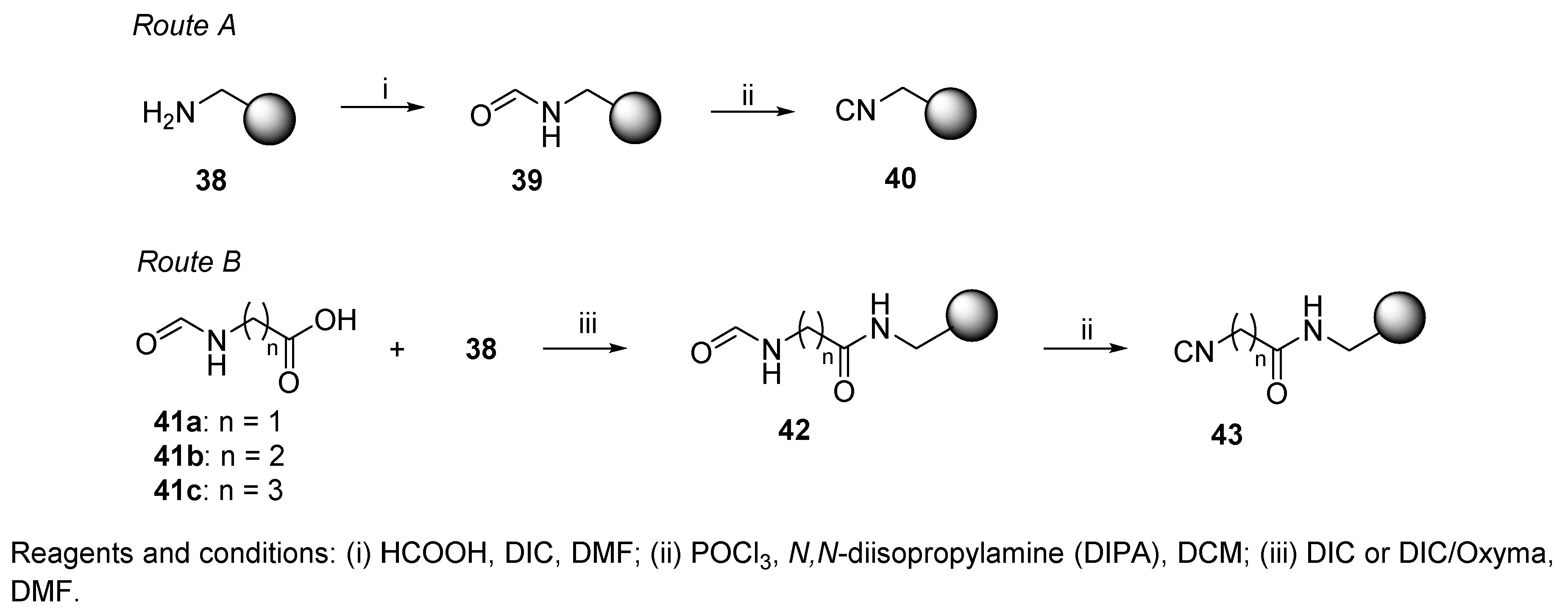
Generation of Hydrogen Cyanide from the Reaction of Oxyma (Ethyl Cyano(hydroxyimino)acetate) and DIC (Diisopropylcarbodiimide) | Organic Process Research & Development

Minimizing HCN in DIC/Oxyma-Mediated Amide Bond-Forming Reactions | Organic Process Research & Development

Use of Oxyma as pH modulatory agent to be used in the prevention of base‐driven side reactions and its effect on 2‐chlorotrityl chloride resin - Subirós‐Funosas - 2012 - Peptide Science -

PDF) Oxyma as cyanooxime building block for the construction of versatile reagents assisting acylation reactions in peptide and organic chemistry
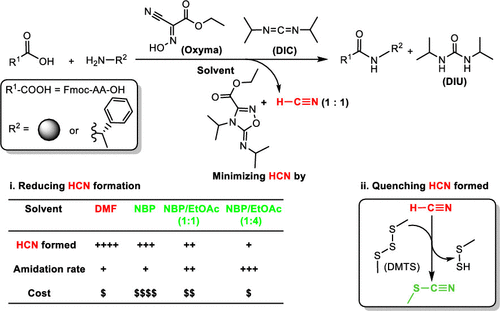
Minimizing HCN in DIC/Oxyma-Mediated Amide Bond-Forming Reactions,Organic Process Research & Development - X-MOL
![Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/0dbaed69-836e-41f8-8db8-526b3a6e9531/mcontent.jpg)
Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library
IJMS | Free Full-Text | Isocyanide Multicomponent Reactions on Solid Phase: State of the Art and Future Application | HTML

Generation of Hydrogen Cyanide from the Reaction of Oxyma (Ethyl Cyano(hydroxyimino)acetate) and DIC (Diisopropylcarbodiimide),Organic Process Research & Development - X-MOL

Prevention of aspartimide formation during peptide synthesis using cyanosulfurylides as carboxylic acid-protecting groups | Nature Communications
![Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/f25812e6-46f3-48f3-8b7c-f247189f2dfd/msch002.jpg)
Oxyma: An Efficient Additive for Peptide Synthesis to Replace the Benzotriazole‐Based HOBt and HOAt with a Lower Risk of Explosion[1] - Subirós‐Funosas - 2009 - Chemistry – A European Journal - Wiley Online Library

Share share 中文 Chinese Journal of Organic Chemistry Quick search Citation search Site search Adv search Toggle navigation CJOG About About Chinese Journal of Organic Chemistry Editorial board Impact factor Indexed in Browse Just accepted Current ...
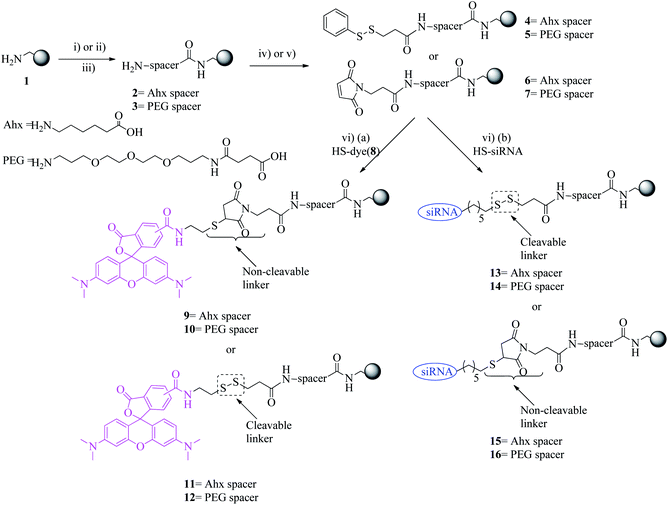
Sulfhydryl reactive microspheres for the efficient delivery of thiolated bioactive cargoes - Journal of Materials Chemistry (RSC Publishing) DOI:10.1039/C1JM11948F