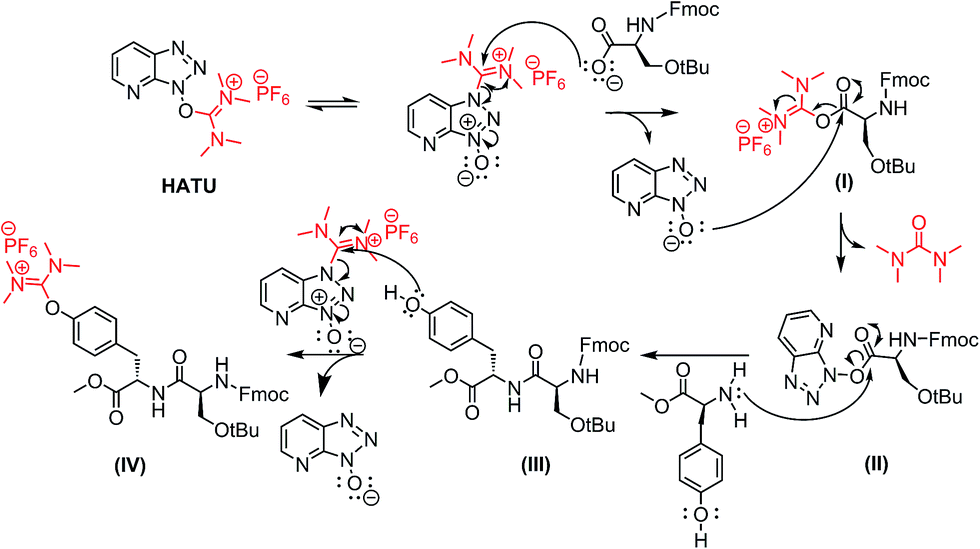
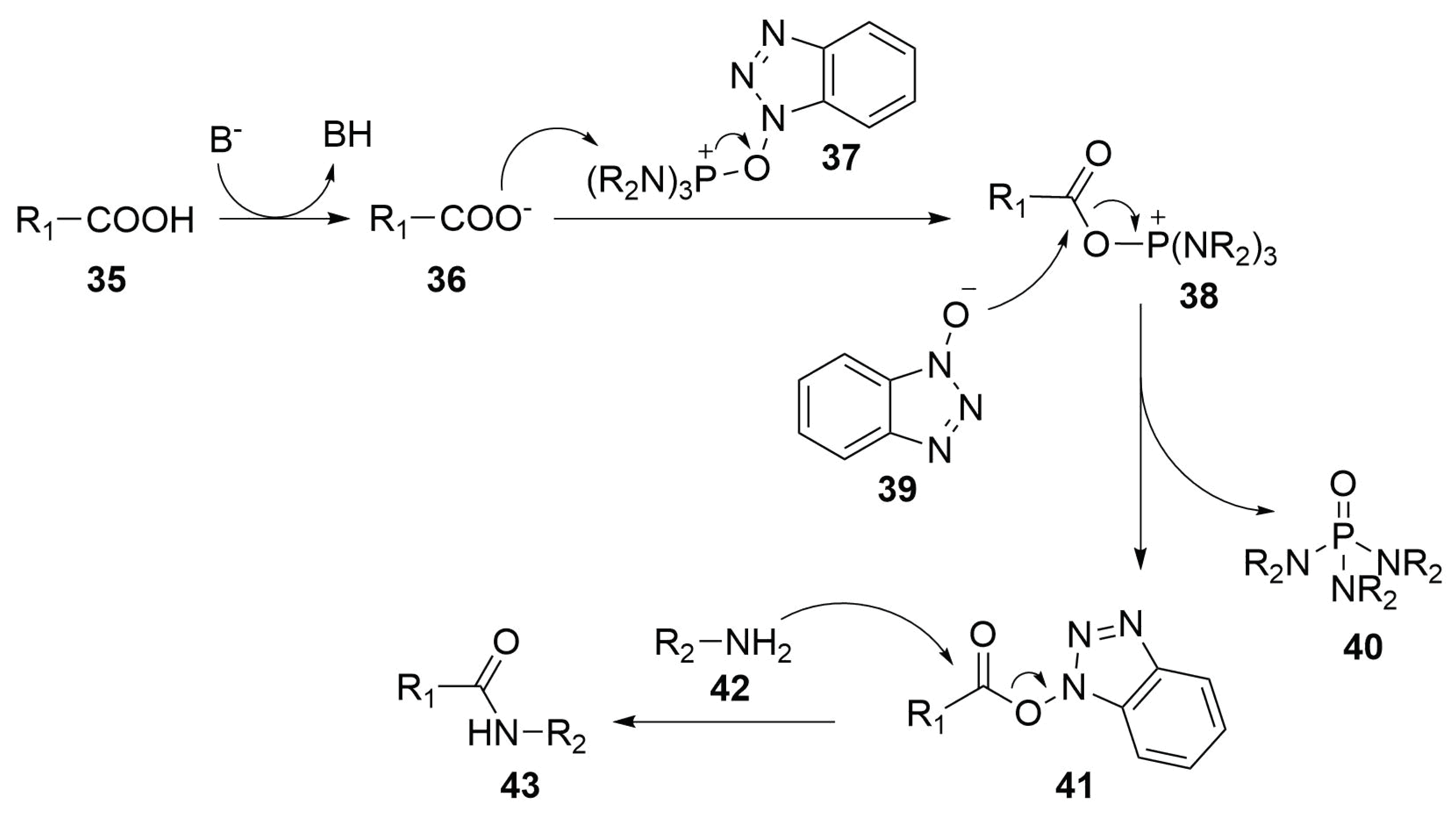
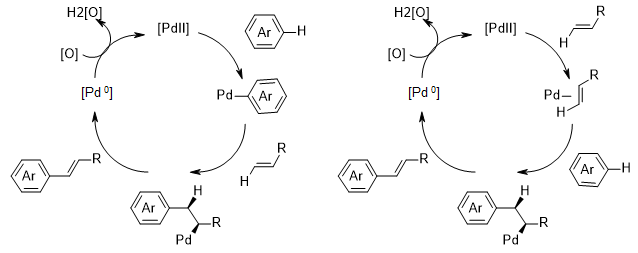
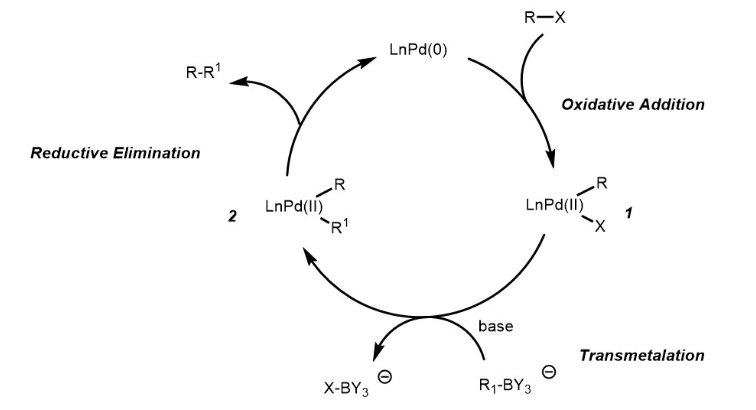
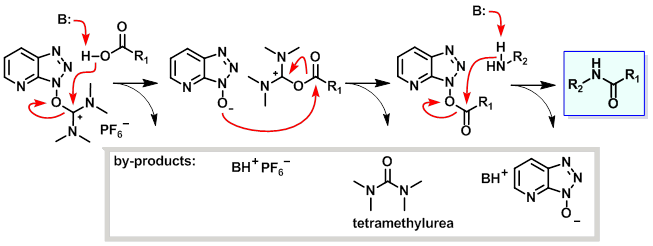
Optimization of Manganese Coupling Reaction for Kilogram-Scale Preparation of Two Aryl-1,3-dione Building Blocks | Organic Process Research & Development
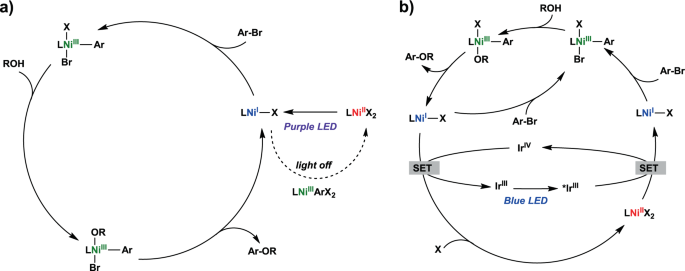
Deciphering the mechanism of the Ni-photocatalyzed C‒O cross-coupling reaction using a tridentate pyridinophane ligand | Nature Communications

Ni-catalyzed deaminative cross-electrophile coupling of Katritzky salts with halides via C─N bond activation
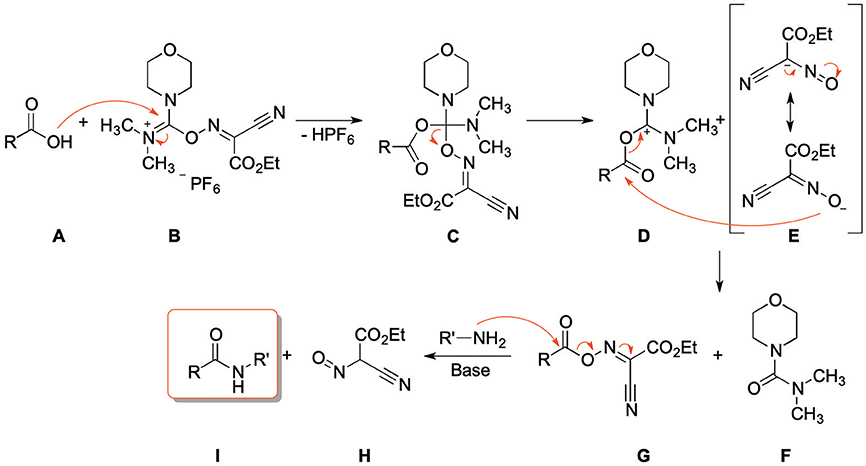
Frontiers | Fast, Efficient, and Versatile Synthesis of 6-amino-5-carboxamidouracils as Precursors for 8-Substituted Xanthines | Chemistry