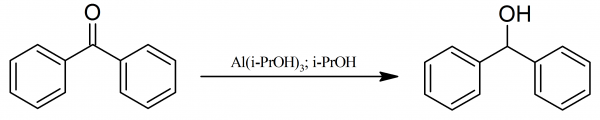EP0033585A1 - A process for the preparation of a benzhydrol derivative and a novel intermediate for use therein - Google Patents

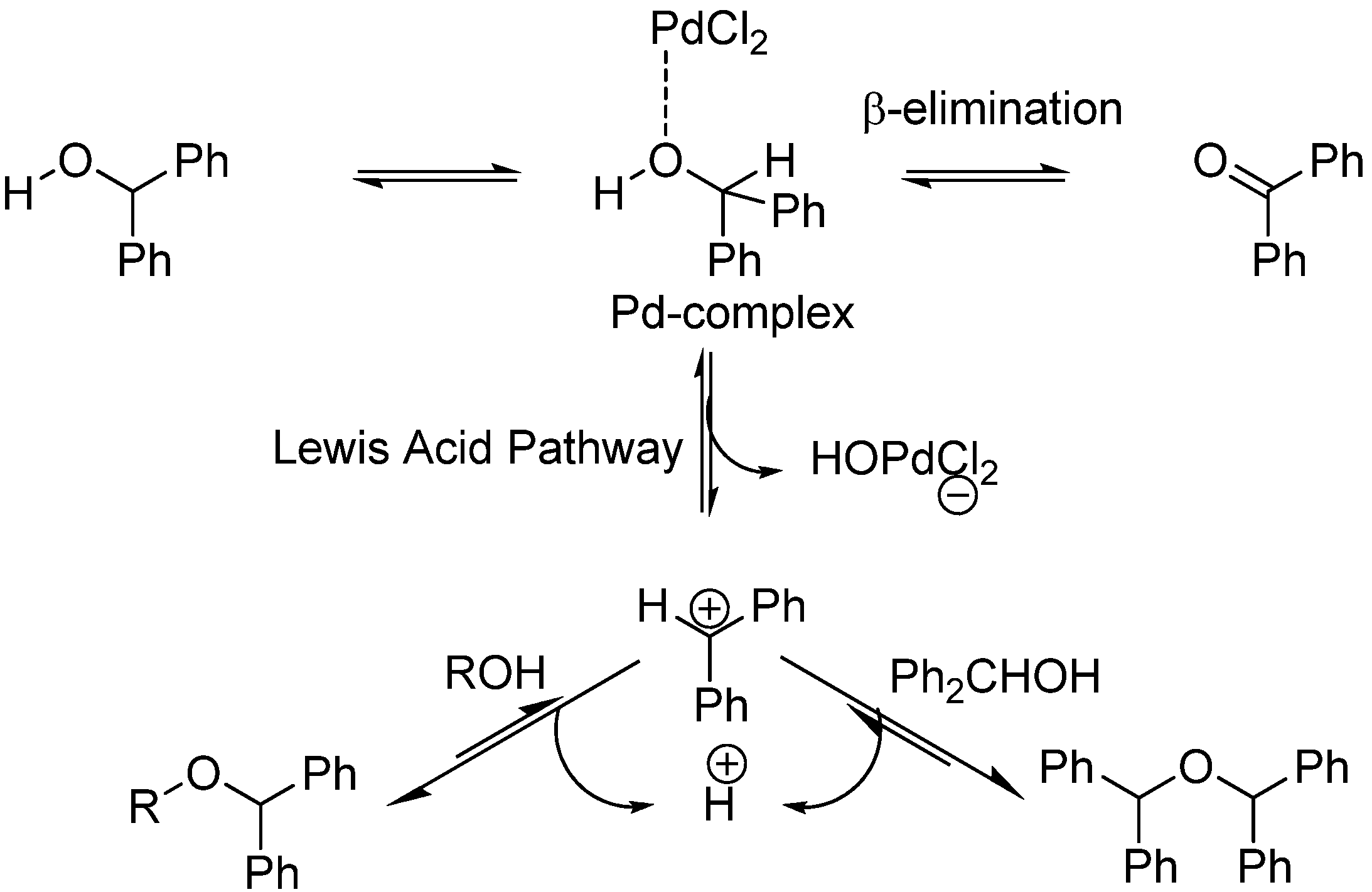
Reaction of an Al/P-based frustrated Lewis pair with benzophenone: formation of adducts and aluminium alcoholates via β-hydride elimination

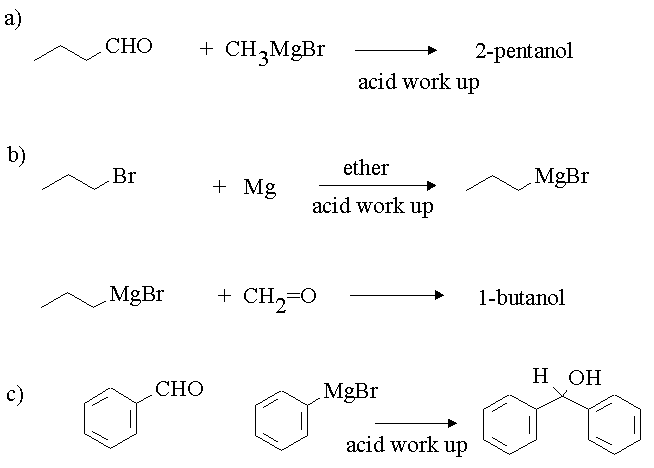
Is this Grignard reaction mechanism to form diphenylmethanol correct? We haven't covered Grignard reactions in theoretical classes yet... : r/chemhelp
Molecules | Free Full-Text | A Straightforward Diphenylmethyl Protection Method and Deprotection of Some Pyrimidine Nucleosides | HTML
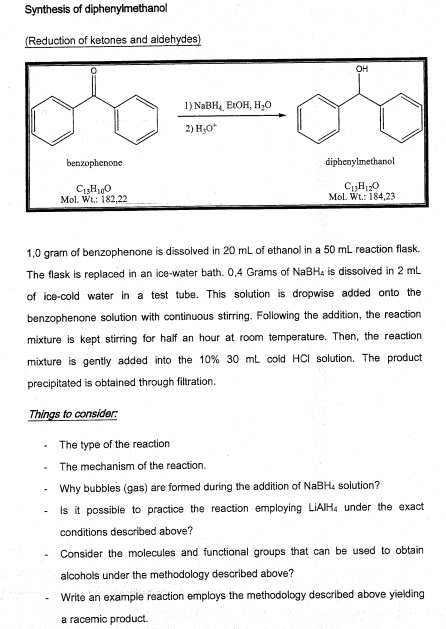
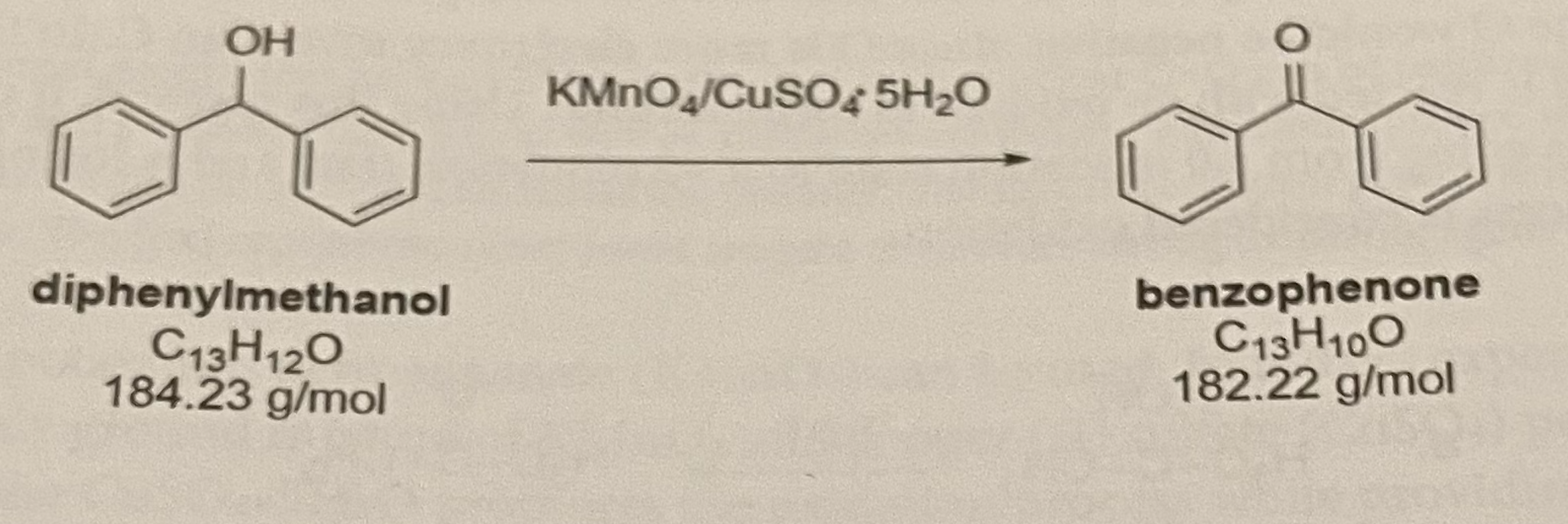
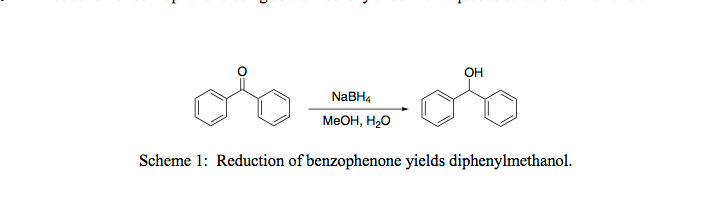
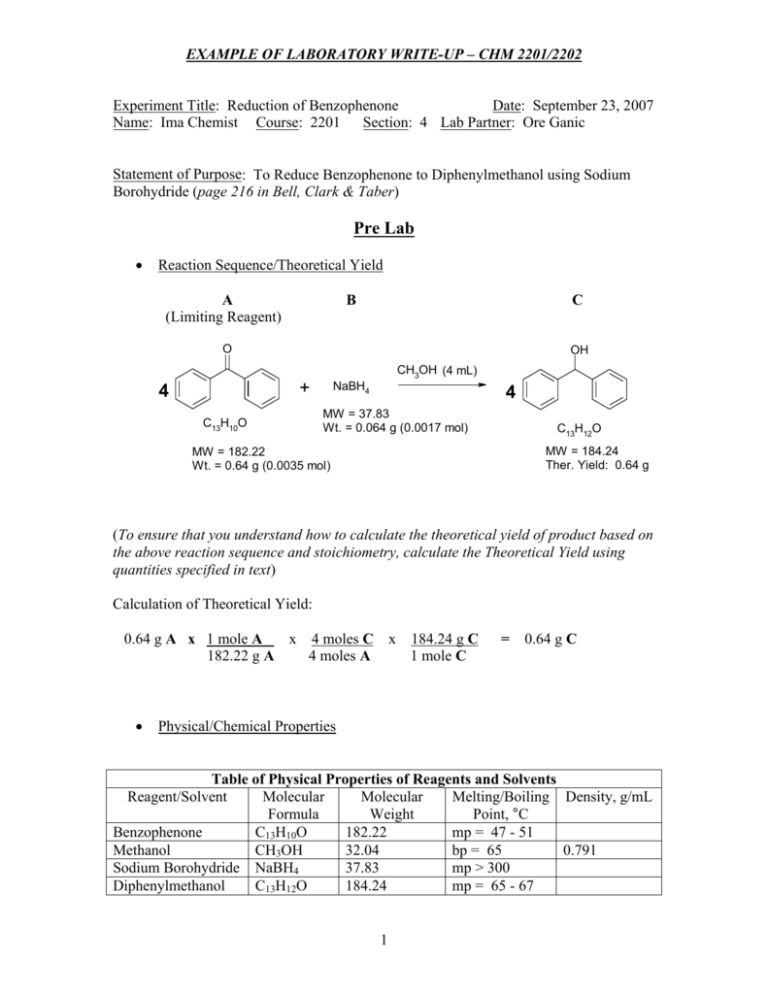
SOLVED:Synthesis of diphenylmethanol Reducten ketones and Idehxdes) 1) NaBl EtOH, H,o 2) Hjot hentubnanona dipbcovimathanol CuHjo MoL CwE,84,23 Mol Wt; 182,22 1,0 gram benzophenone d ssolved 20 ML ethano 50 mL reaction

Exploring the mechanism of diphenylmethanol oxidation: A combined experimental and theoretical approach - ScienceDirect

OneClass: Benzilic Acid Rearrangement lab: 1.) A side product formed in the benzilic acid rearrangeme...

Oxidation of benzhydrol by tributylammonium chlorochromate: a kinetic and mechanistic study | SpringerLink

Role of TiO2-based photocatalysts on the synthesis of the pharmaceutical precursor benzhydrol by UVA-LED radiation - ScienceDirect

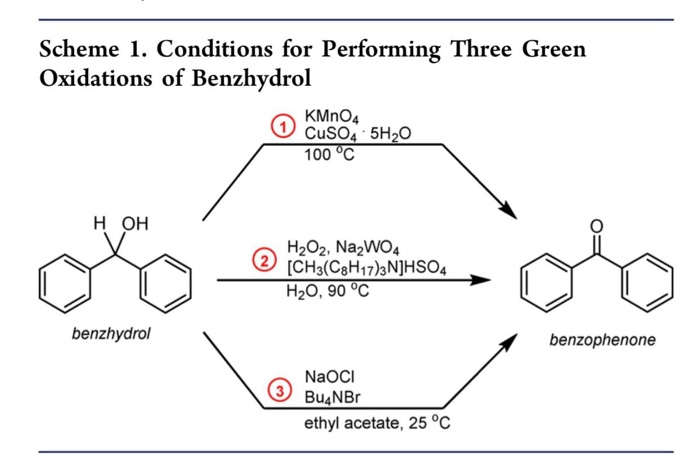
Scheme 3 | Structural and catalytic properties of V-SBA-15 for the vapor phase oxidation of diphenylmethane | SpringerLink