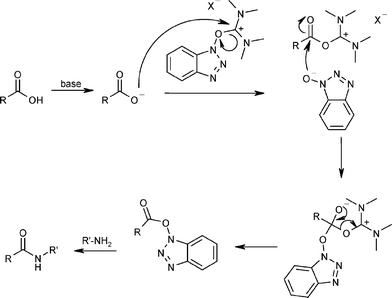
Molecules | Free Full-Text | TOMBU and COMBU as Novel Uronium-Type Peptide Coupling Reagents Derived from Oxyma-B | HTML
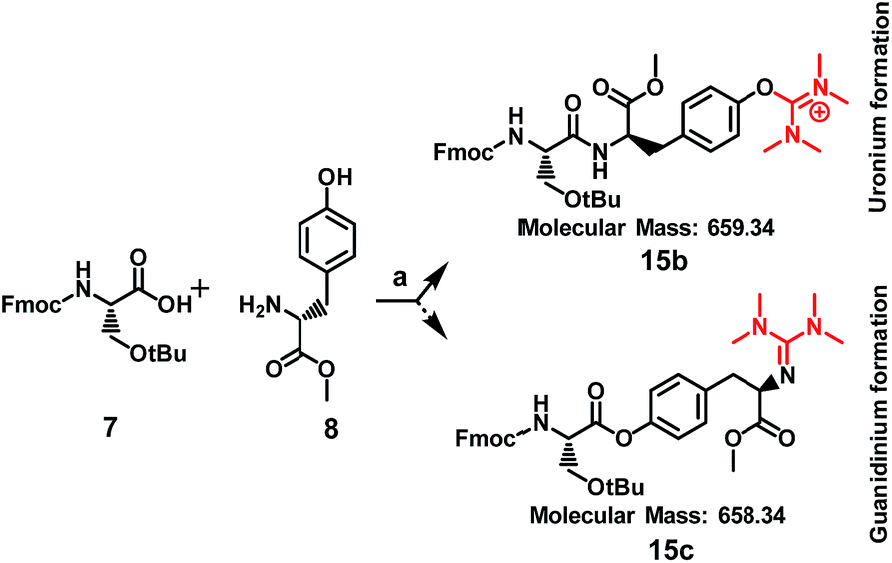
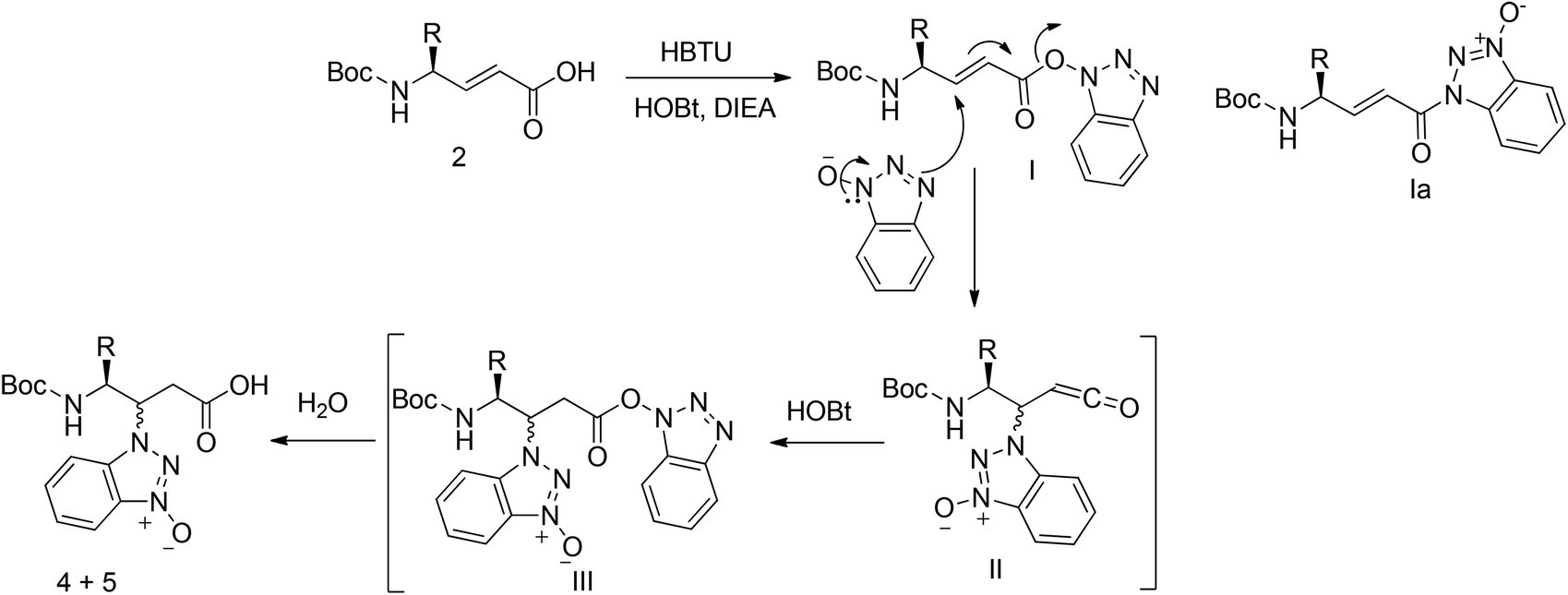
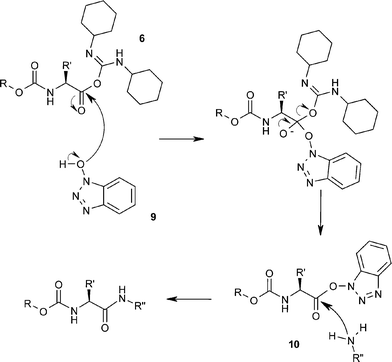
Unveiling and tackling guanidinium peptide coupling reagent side reactions towards the development of peptide-drug conjugates - RSC Advances (RSC Publishing) DOI:10.1039/C7RA06655D

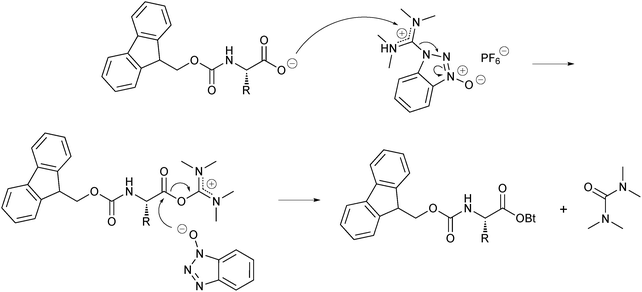
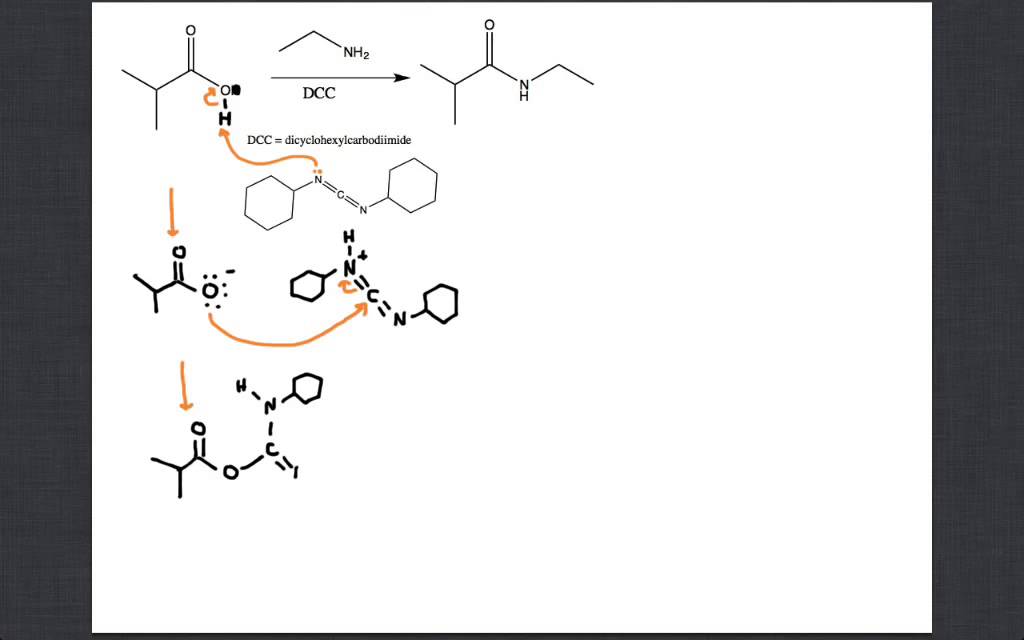
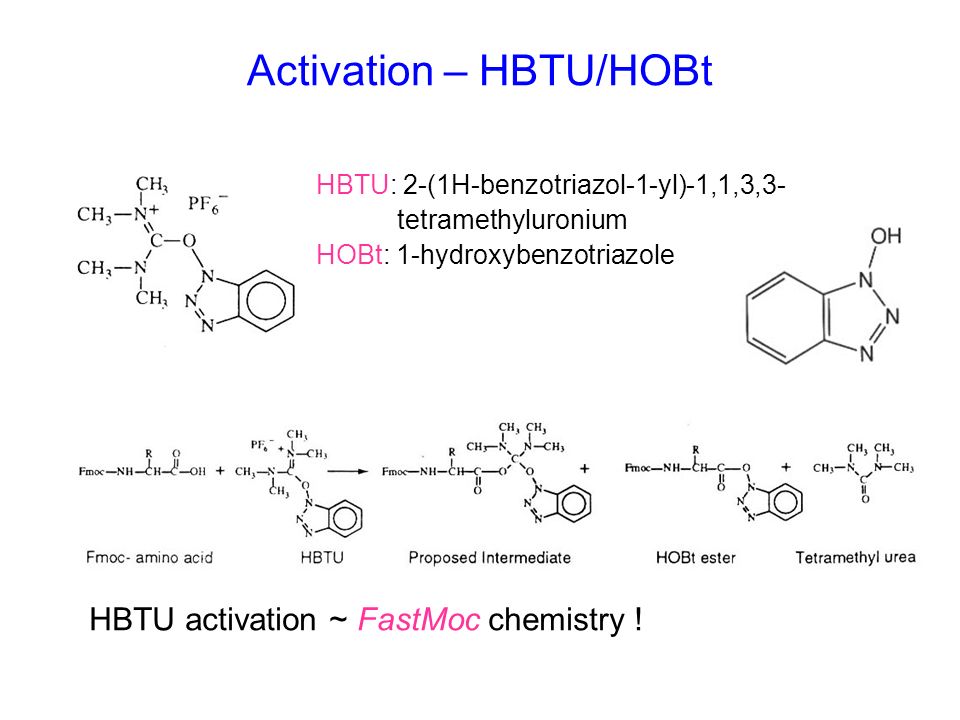
DEPBT as an efficient coupling reagent for amide bond formation with remarkable resistance to racemization - Ye - 2005 - Peptide Science - Wiley Online Library
![The X-ray structures of HOBt-based immonium-type coupling reagents and the rearrangement of benzotriazolyl esters of Nα-protected amino acids or peptides: N- vs. O-substituted forms [ ] - Journal of the Chemical Society, The X-ray structures of HOBt-based immonium-type coupling reagents and the rearrangement of benzotriazolyl esters of Nα-protected amino acids or peptides: N- vs. O-substituted forms [ ] - Journal of the Chemical Society,](https://pubs.rsc.org/image/article/2001/p2/b003930f/b003930f-f2.gif)