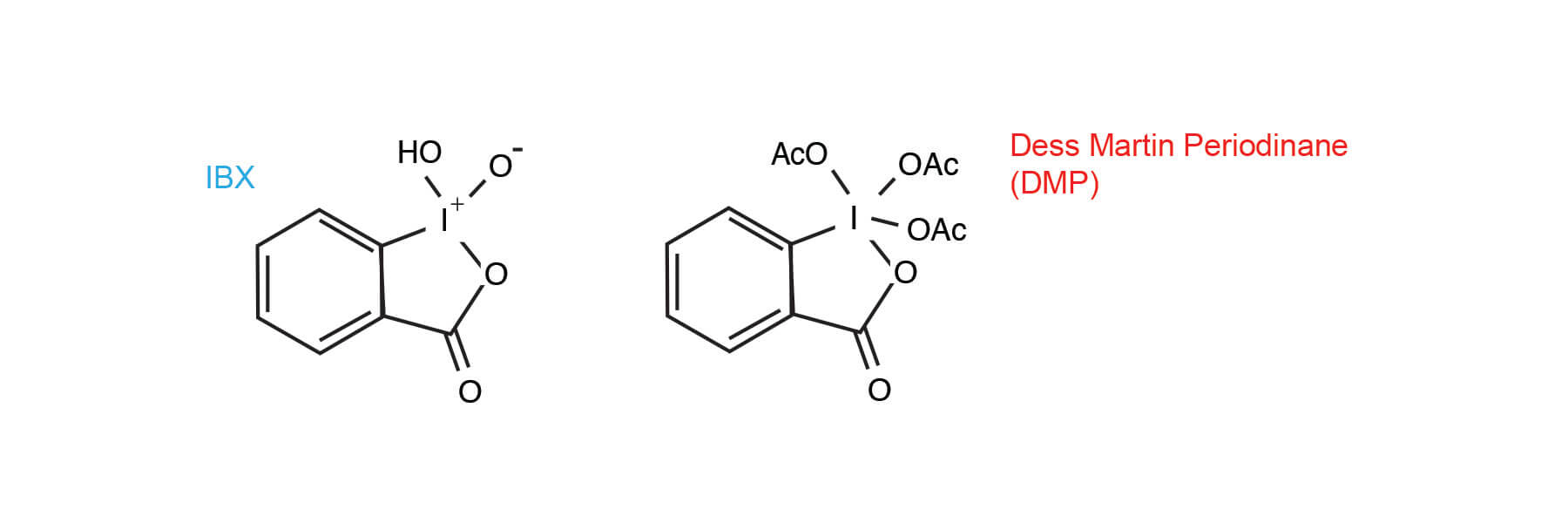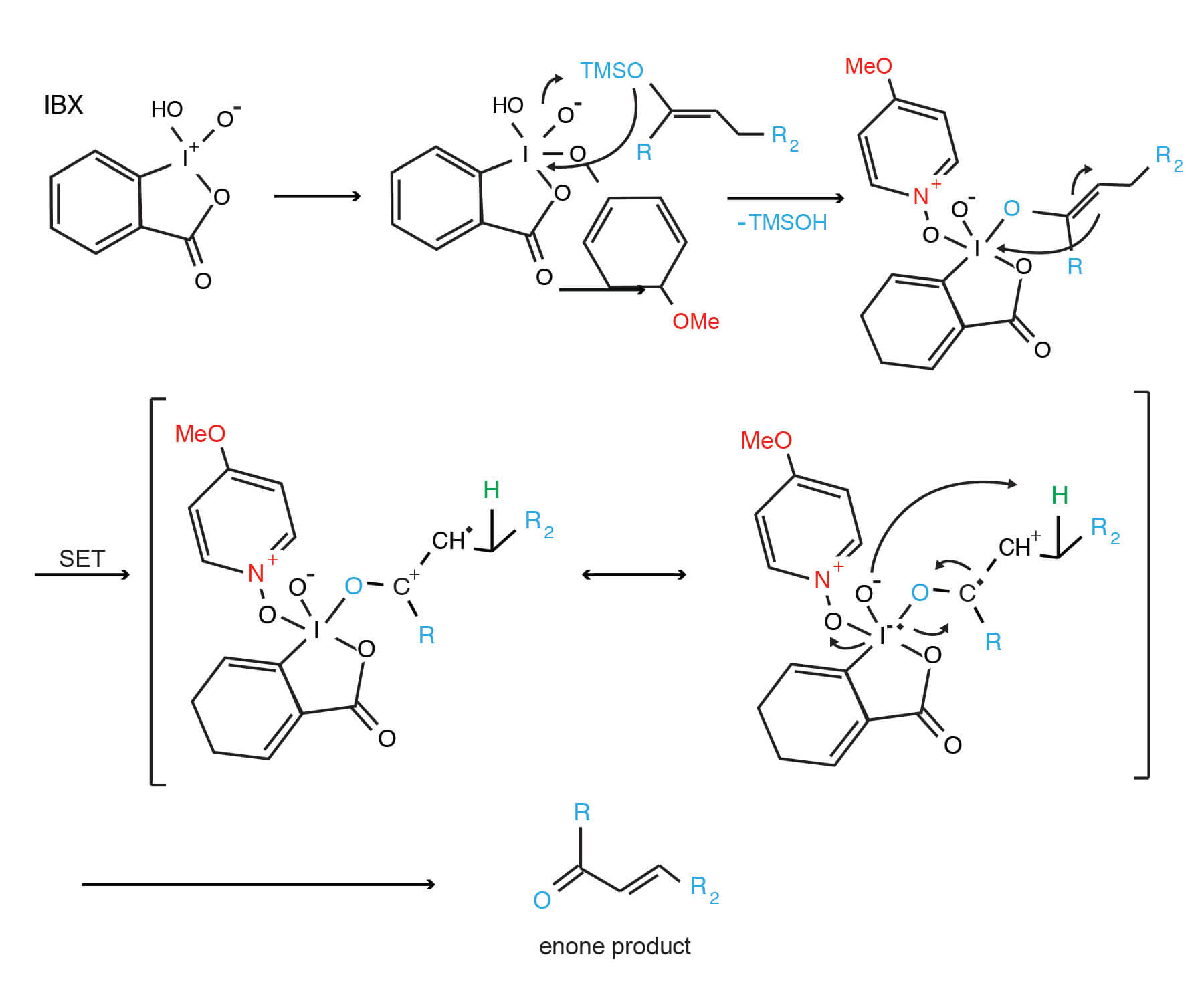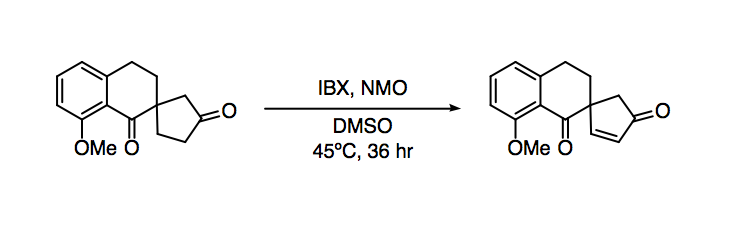
Report: Oxidation of Alkenes in Aqueous Solvent Mixtures Using Environmentally Benign Reagents (58th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
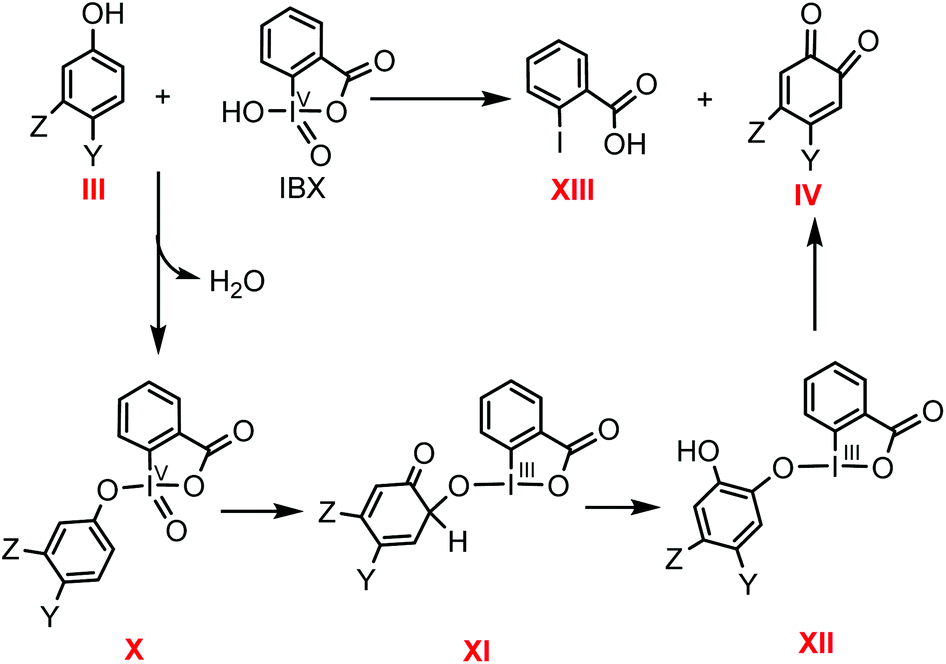
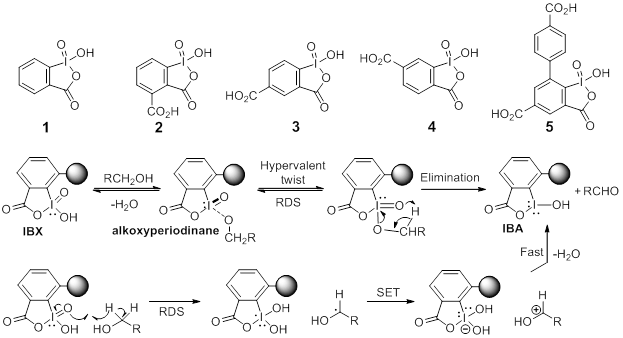
Mechanistic investigation into phenol oxidation by IBX elucidated by DFT calculations - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB02650A
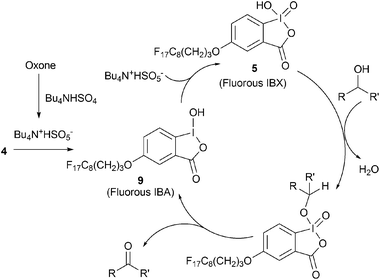
An effective and catalytic oxidation using recyclable fluorous IBX - Chemical Communications (RSC Publishing) DOI:10.1039/C0CC03149F

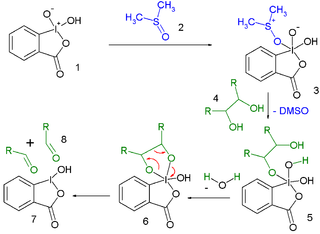
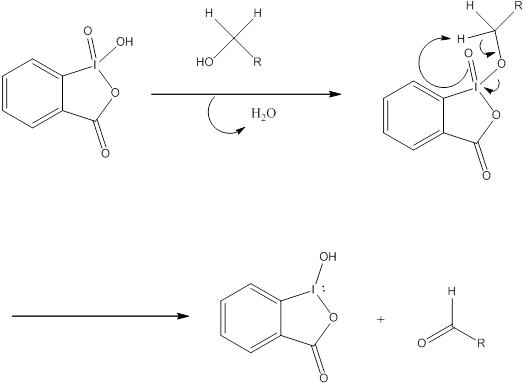
Scheme 4. General mechanism of oxidation of primary alcohols with IBX. | Download Scientific Diagram

Scheme 3 | Functional group transformation from amines to aldehydes via IBX oxidation | SpringerLink
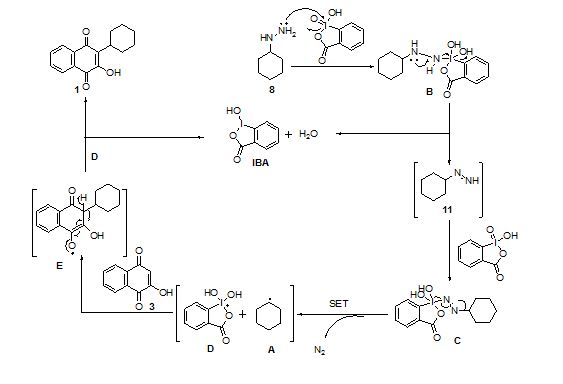
GUEST BLOGGER, Dr Pravin Patil, A New Combination of Cyclohexylhydrazine and IBX for Oxidative Generation of Cyclohexyl Free Radical and Related Synthesis of Parvaquone – All About Drugs

New Reactions of IBX: Oxidation of Nitrogen‐ and Sulfur‐Containing Substrates To Afford Useful Synthetic Intermediates - Nicolaou - 2003 - Angewandte Chemie - Wiley Online Library

Selective oxidation of benzylic carbons to the corresponding carbonyl groups using IBX and Oxone in user and eco-friendly solvents