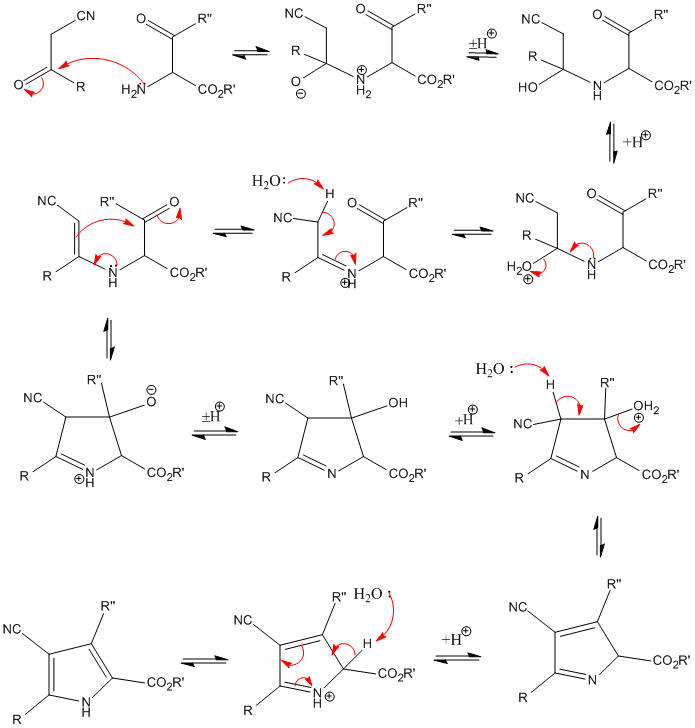
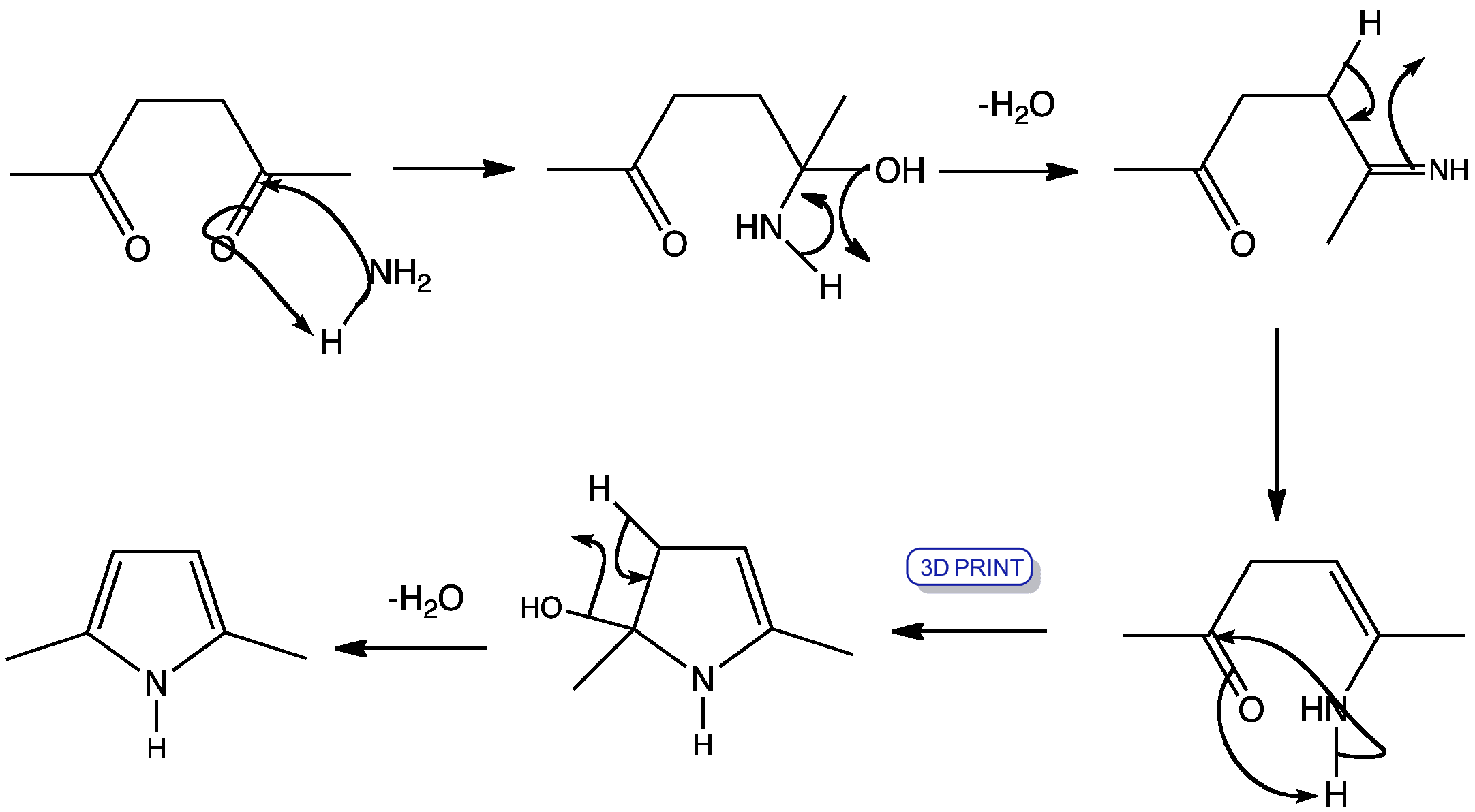
Mechanism of the Paal–Knorr reaction: the importance of water mediated hemialcohol pathway - RSC Advances (RSC Publishing) DOI:10.1039/C5RA16246G
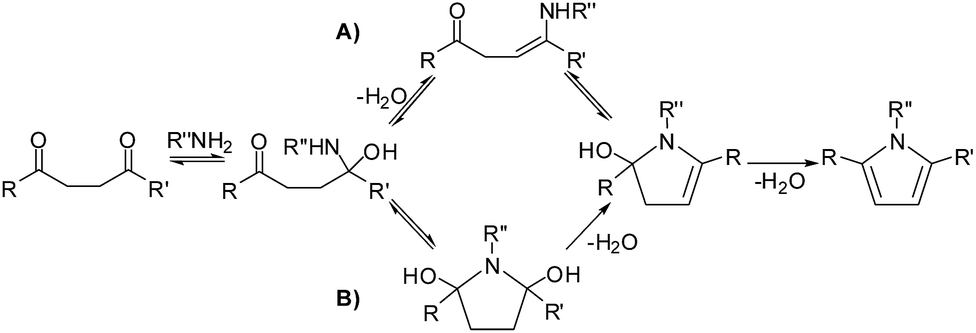
Mechanism of the Paal–Knorr reaction: the importance of water mediated hemialcohol pathway - RSC Advances (RSC Publishing) DOI:10.1039/C5RA16246G
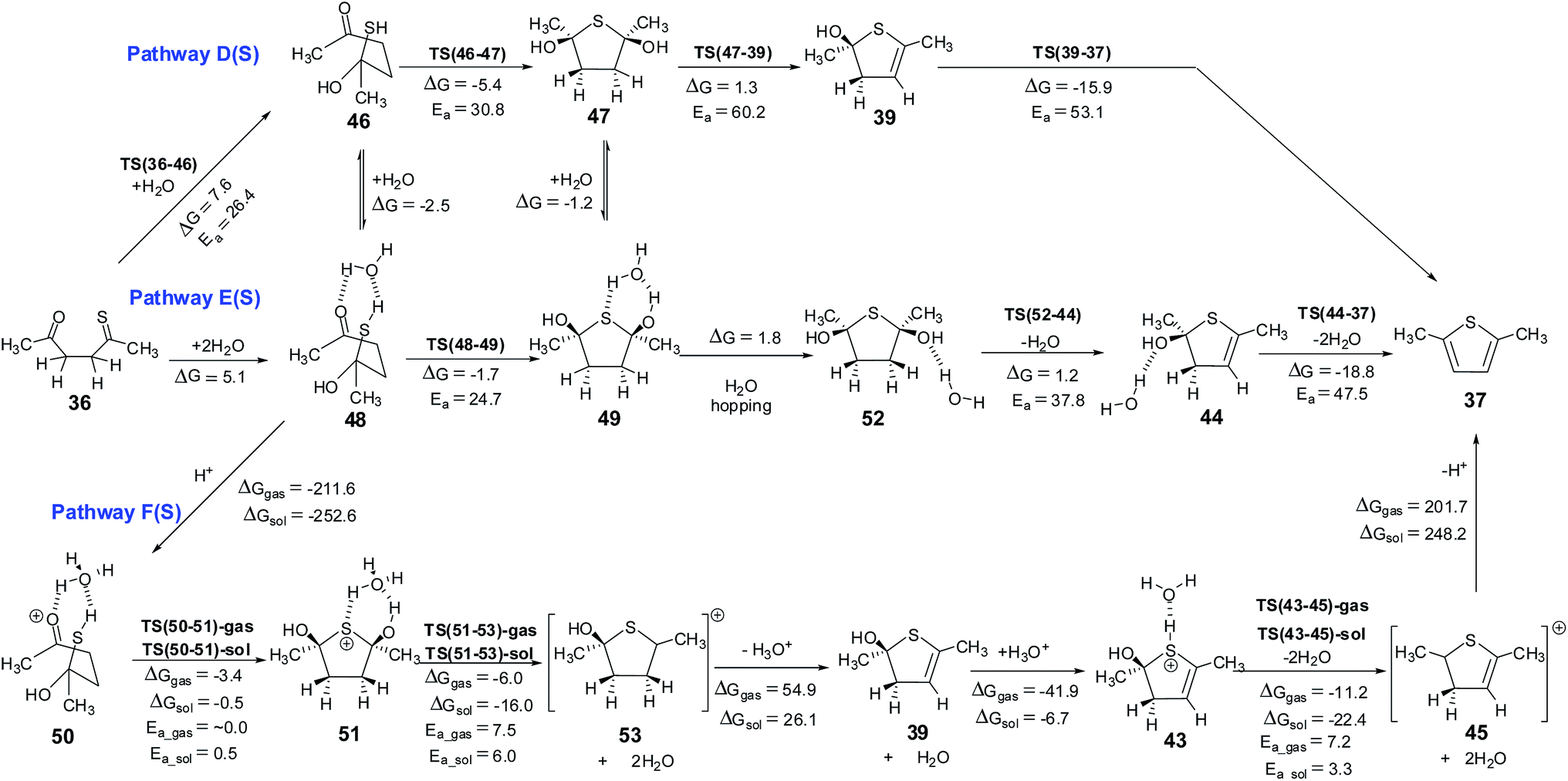
Mechanism of the Paal–Knorr reaction: the importance of water mediated hemialcohol pathway - RSC Advances (RSC Publishing) DOI:10.1039/C5RA16246G
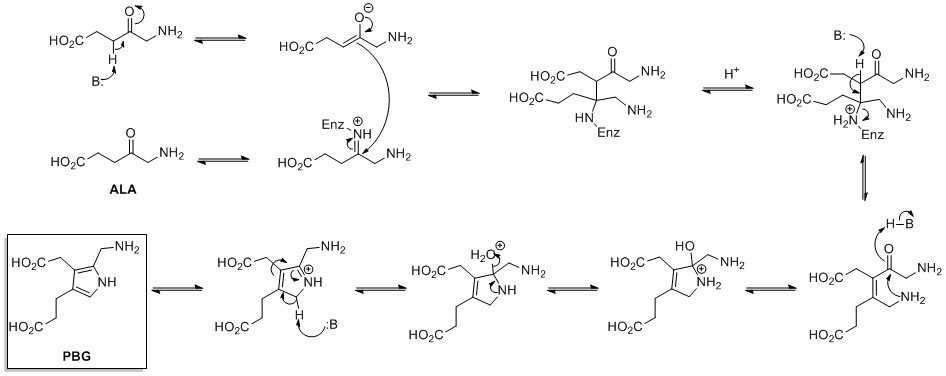
![Molecules | Free Full-Text | Synthesis of Multi-Substituted Pyrrole Derivatives Through [3+2] Cycloaddition with Tosylmethyl Isocyanides (TosMICs) and Electron-Deficient Compounds | HTML Molecules | Free Full-Text | Synthesis of Multi-Substituted Pyrrole Derivatives Through [3+2] Cycloaddition with Tosylmethyl Isocyanides (TosMICs) and Electron-Deficient Compounds | HTML](https://www.mdpi.com/molecules/molecules-23-02666/article_deploy/html/images/molecules-23-02666-sch001.png)
Molecules | Free Full-Text | Synthesis of Multi-Substituted Pyrrole Derivatives Through [3+2] Cycloaddition with Tosylmethyl Isocyanides (TosMICs) and Electron-Deficient Compounds | HTML

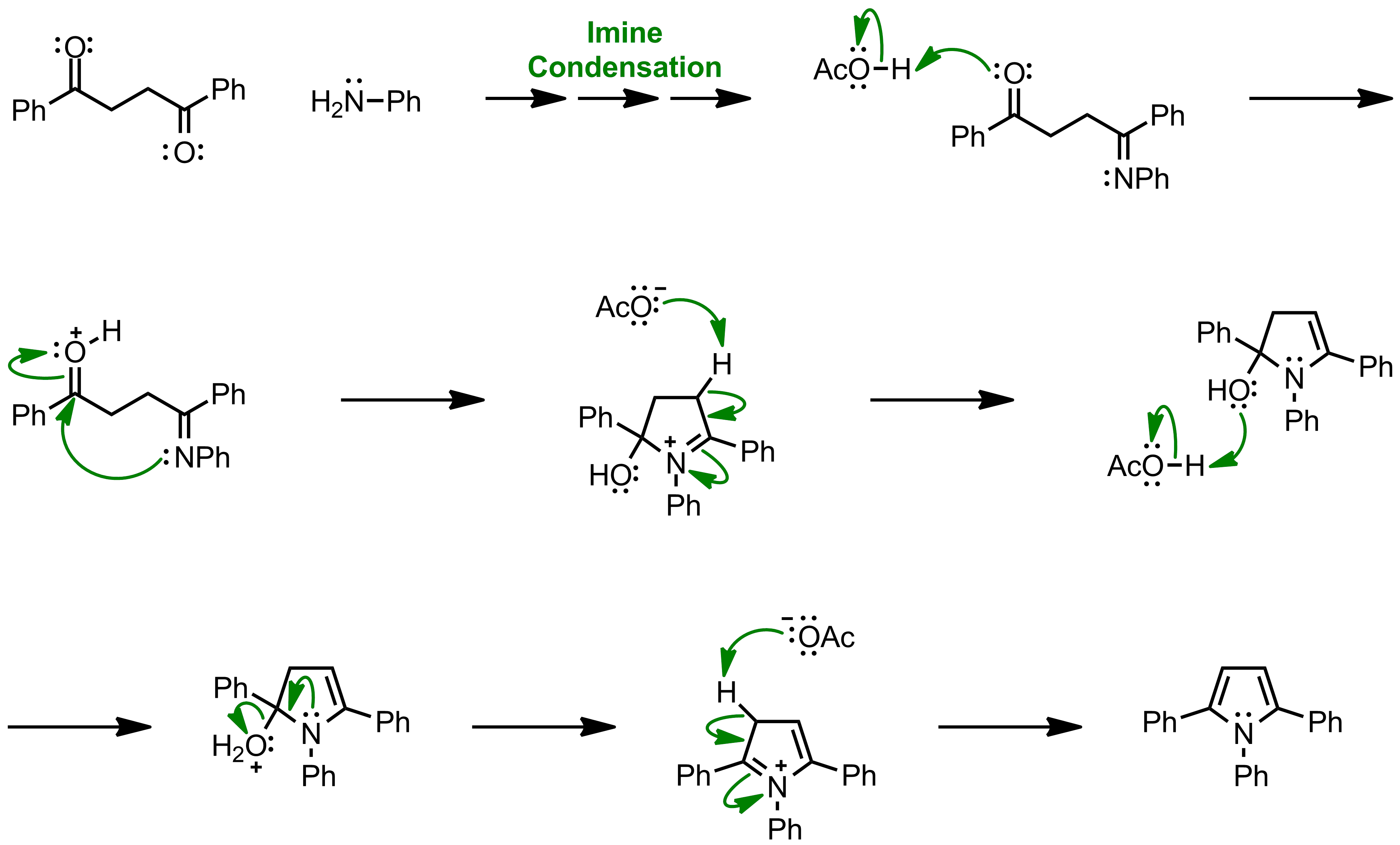
An approach to the Paal–Knorr pyrroles synthesis in the presence of β-cyclodextrin in aqueous media - ScienceDirect

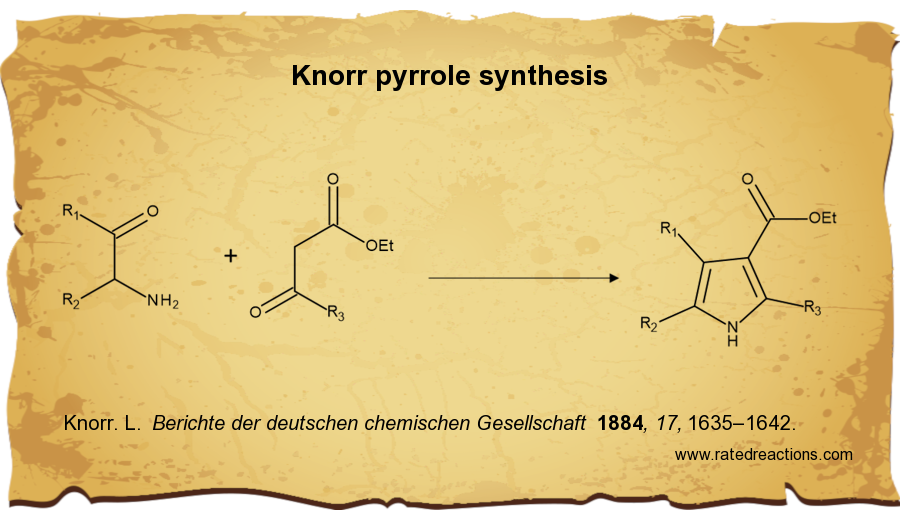
Paal-Knorr furan, thiophene, pyrrole synthesis made by Roman A. Valiulin with ChemDraw | Organic chemistry, Writing words, Organic chemistry reactions




.jpg)