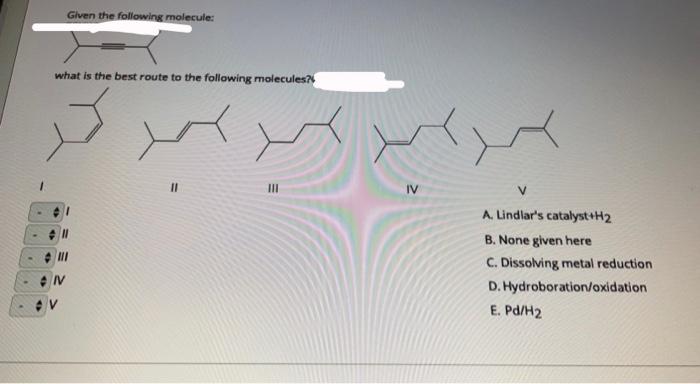
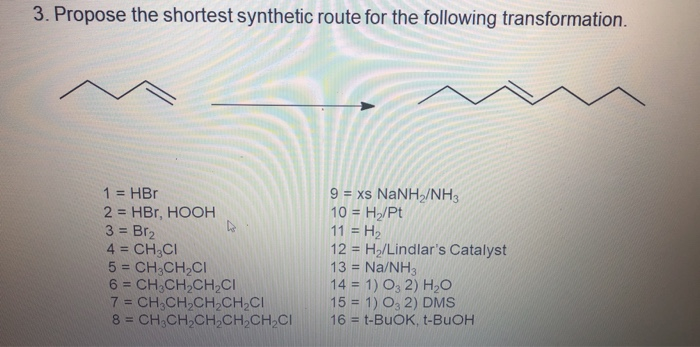
SOLVED:Propose the shortest synthetic route for the following transformation Select reagents from following for the multistep synthesis 1 =HBr aX NaNH /NH, 2 = HBr; HOOH 10 = H,/Pt 3 = 8r2

SOLVED:Detse synthetic route - the did shown below starting with 1,1,3,3-tetramethyl-2-ethylcydohexa 1) Brz hv; 2) KO-+-Bu; HBr; RCOR; Ko-t-Bu; 8rz, CCl; 6) xs NaNHz; 7) Hzo; HCI; 8rz, hv; 2) KO-+Bu; 3)
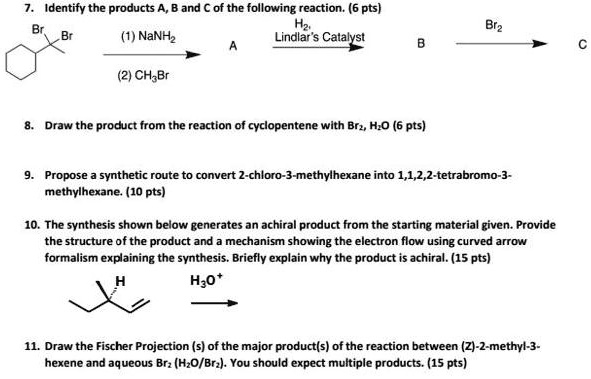
SOLVED:Identify the products and of the following reaction. (6 pts) NaNHz Lindlar'$ Catalyst (2) CH-Br Draw the product from the reaction of cyclopentene with Brz, HzO (6 pts) Propose synthetic route to

From the Lindlar Catalyst to Supported Ligand‐Modified Palladium Nanoparticles: Selectivity Patterns and Accessibility Constraints in the Continuous‐Flow Three‐Phase Hydrogenation of Acetylenic Compounds - Vilé - 2014 - Chemistry – A European Journal -