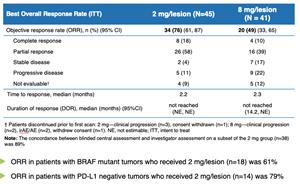
Table 3 from New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). | Semantic Scholar

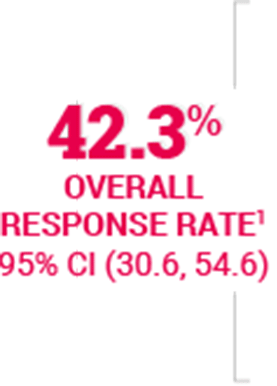
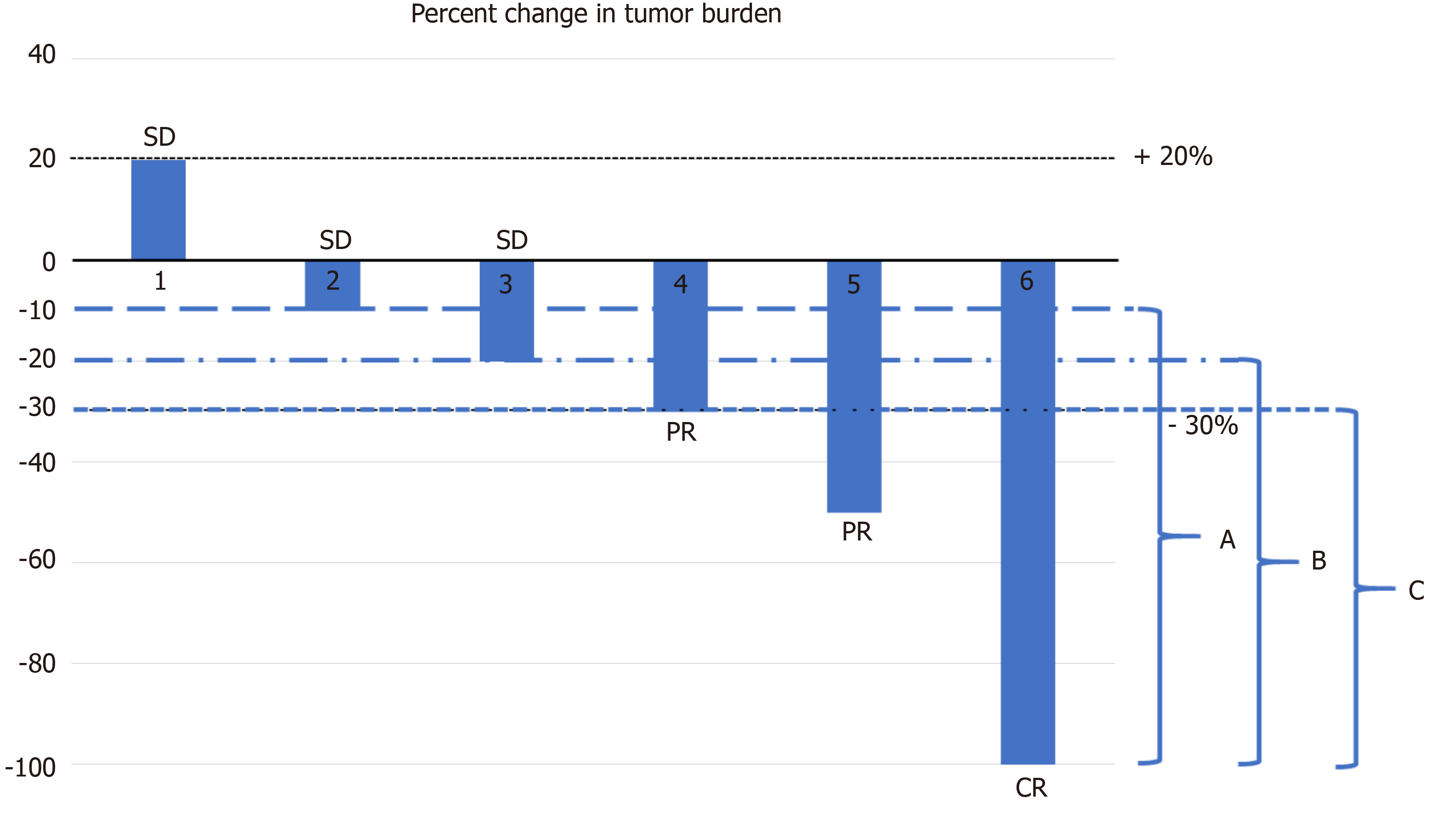
Tumor Response Rates | Advanced NSCLC | ABRAXANE (paclitaxel protein-bound particles for injectable suspension) (albuminbound)