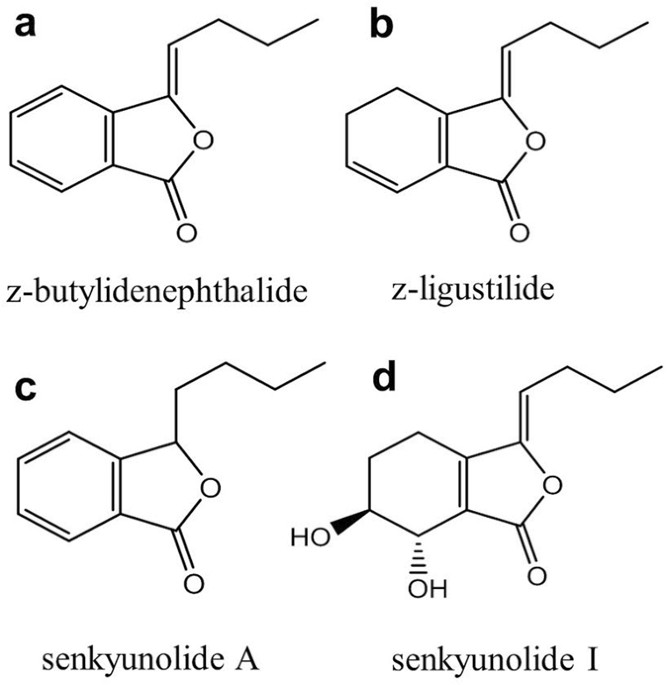
Phthalide Derivatives from Angelica Sinensis Decrease Hemoglobin Oxygen Affinity: A New Allosteric-Modulating Mechanism and Potential Use as 2,3-BPG Functional Substitutes | Scientific Reports
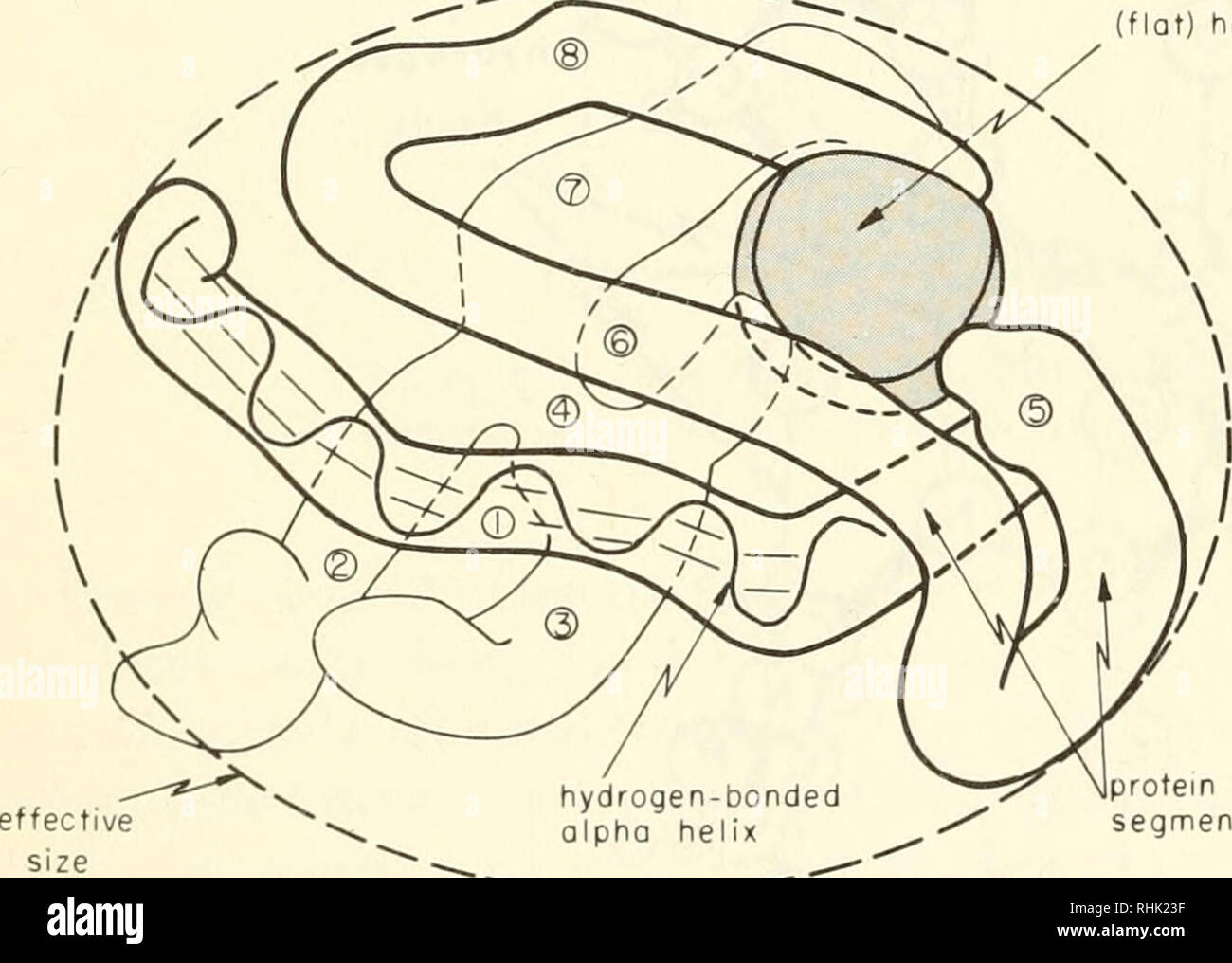
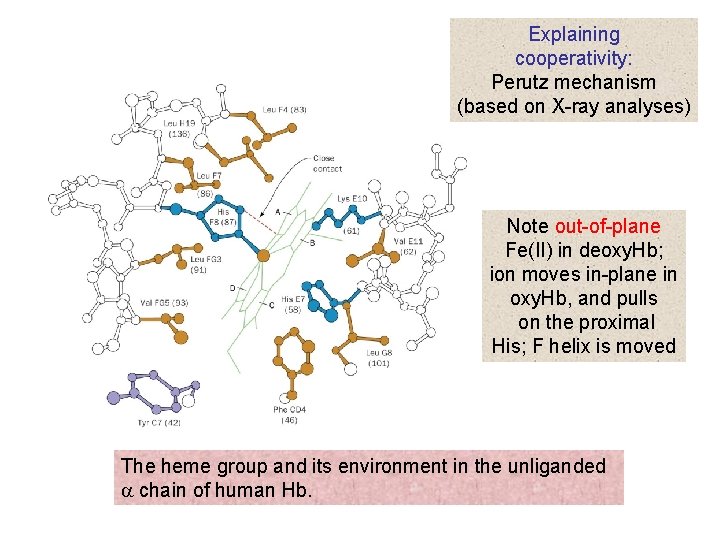
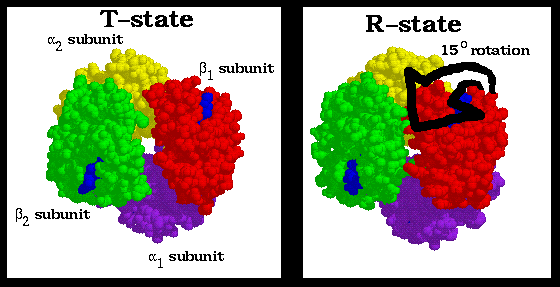
Biophysics: concepts and mechanisms. Biophysics. 132 BIG MOLECULES investigation. The X-ray diffraction pattern of even single crystals was too formidable for analysis until M. F. Perutz, about 1950, began to substitute
Chapter 16 The Molecular Basis of Inheritance. In 1936, Max Perutz, winner of the Nobel Prize in Chemistry in 1962 for his study of hemoglobin, said: - ppt download

Max Perutz Labs - Gang Dong's group has revealed a conserved mechanism for centriole targeting of Plk4 kinases – their recent paper published in Structure was highlighted in the August issue of

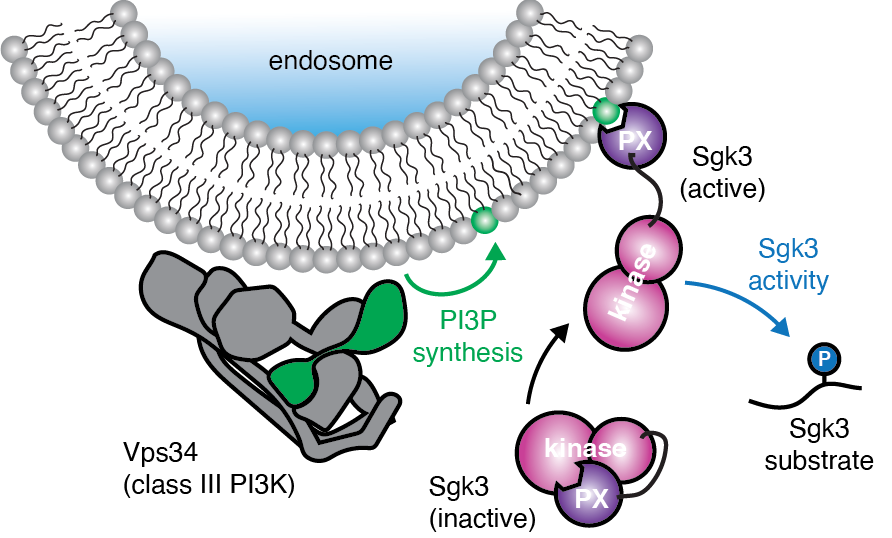
Max Perutz Labs Vienna on Twitter: "The Kuchler lab has unraveled the molecular mechanism of the human ABCG2 drug transporter in their recent paper in @NatureComms. The results suggest new therapeutic strategies

Comparison of the MWC, extended MWC/Perutz, and global allostery models... | Download Scientific Diagram

The framework of RA-CasRel model with a concrete example, where the... | Download Scientific Diagram

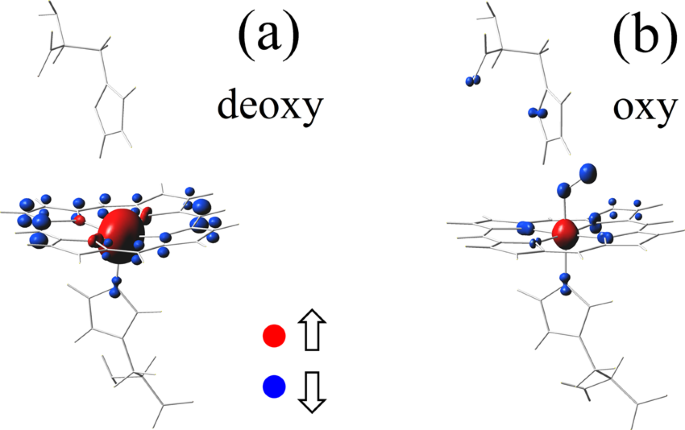
New Insights into the Allosteric Mechanism of Human Hemoglobin from Molecular Dynamics Simulations - ScienceDirect