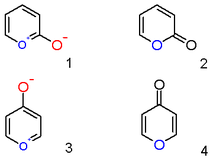
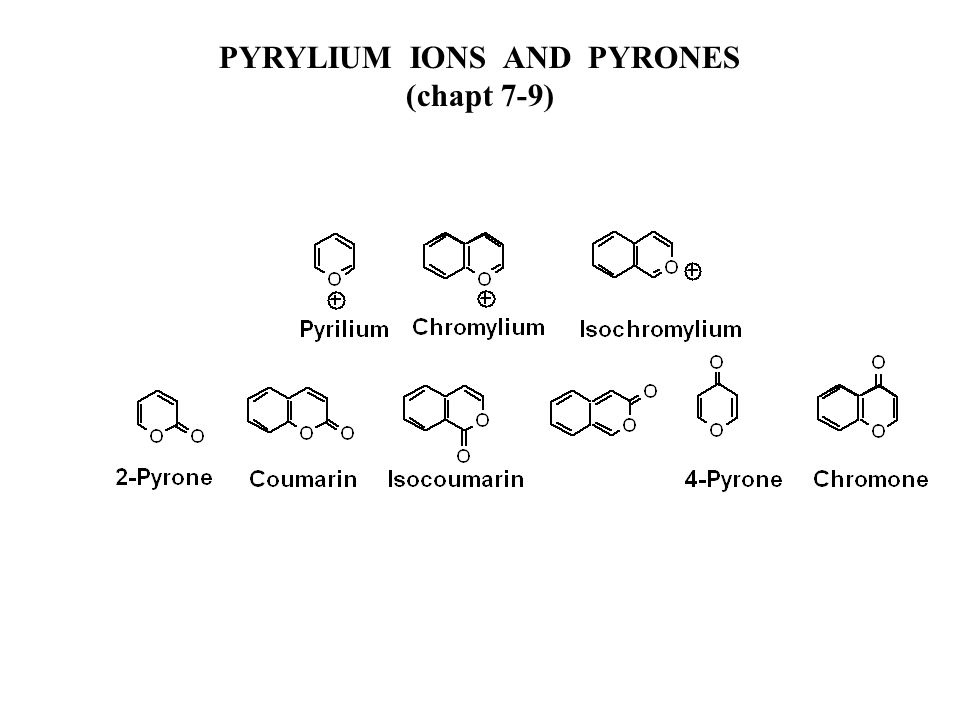
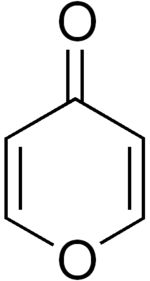
The three most common isomers of a furan ring fused to the α-pyrone... | Download Scientific Diagram

Recent Advances in the Synthesis of 2-Pyrones – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.

α-pyrones: Small molecules with versatile structural diversity reflected in multiple pharmacological activities-an update - ScienceDirect
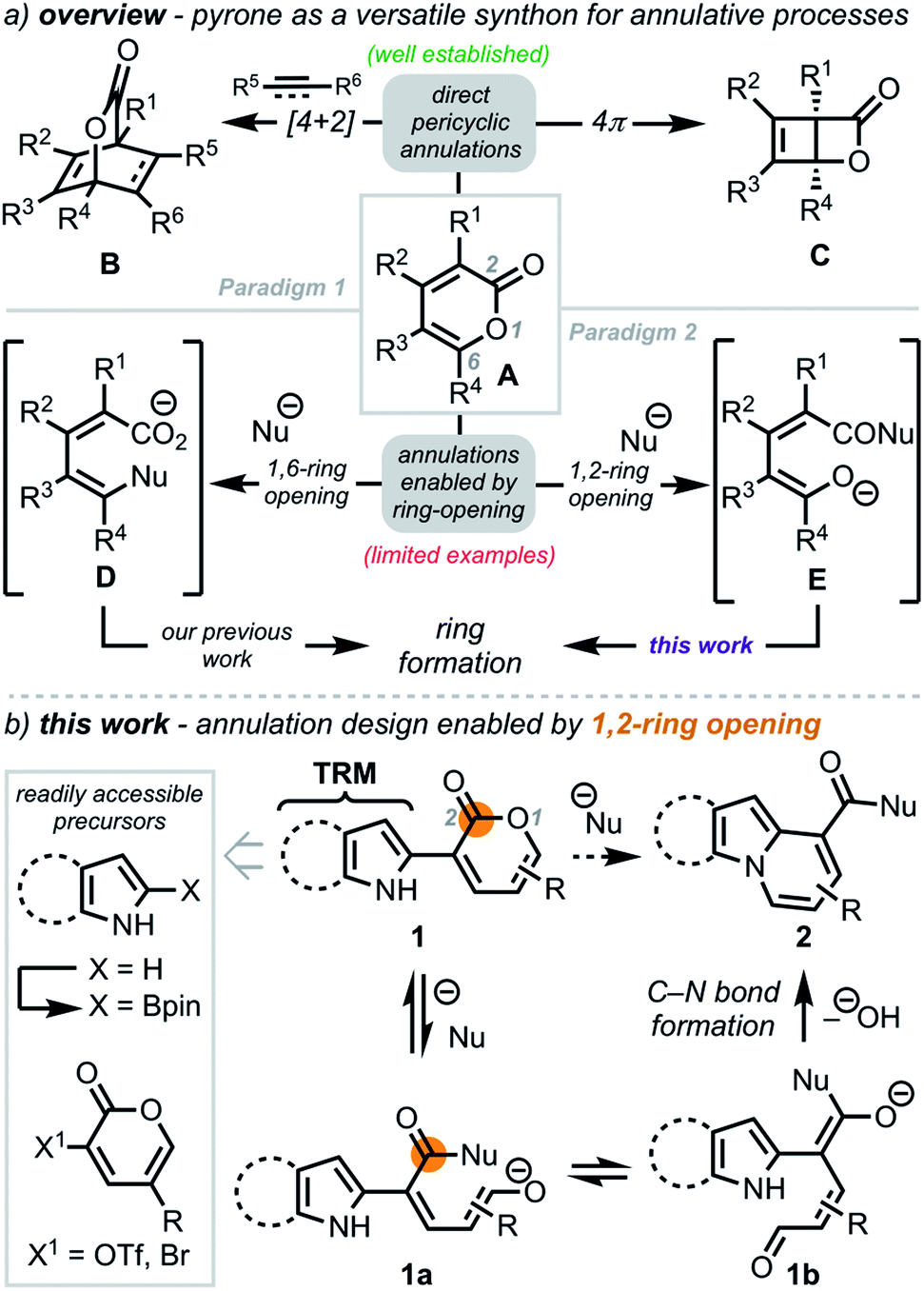
A pyrone remodeling strategy to access diverse heterocycles: application to the synthesis of fascaplysin natural products - Chemical Science (RSC Publishing)

Photoinduced transformation of matrix-isolated methyl 2-pyrone-3-carboxylate into methyl 2-pyrone-5-carboxylate via intramolecular hydrogen shift in open-ring aldehyde–ketene - ScienceDirect
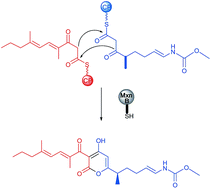
In vitro reconstitution of α-pyrone ring formation in myxopyronin biosynthesis - Chemical Science (RSC Publishing)
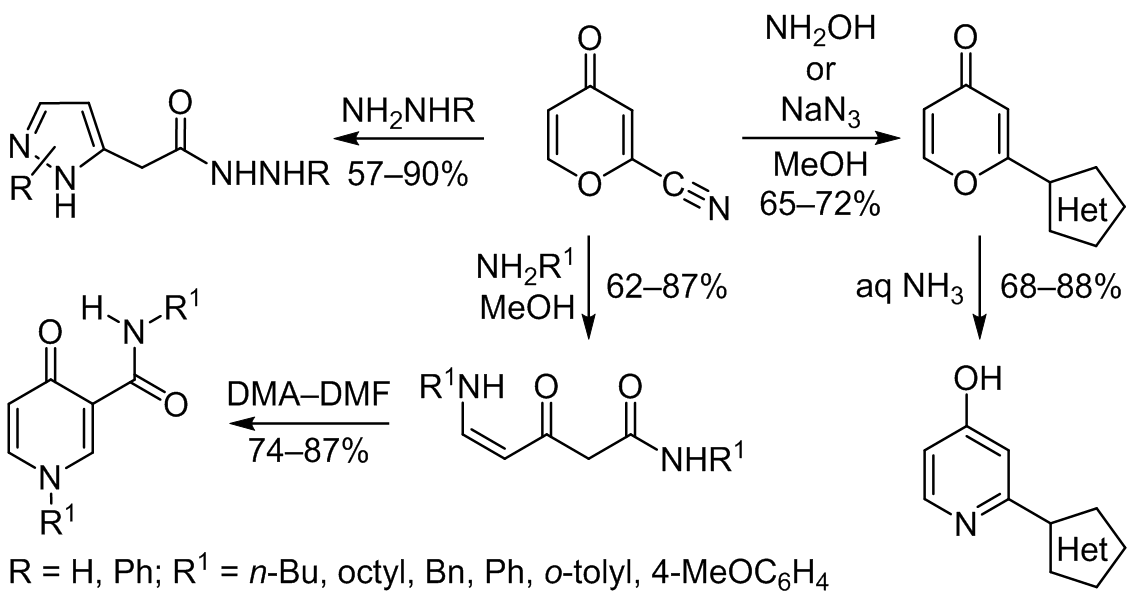
SYNTHESIS AND SOME CHEMICAL PROPERTIES OF 2-CYANO-4-PYRONE | Обыденнов | Chemistry of Heterocyclic Compounds
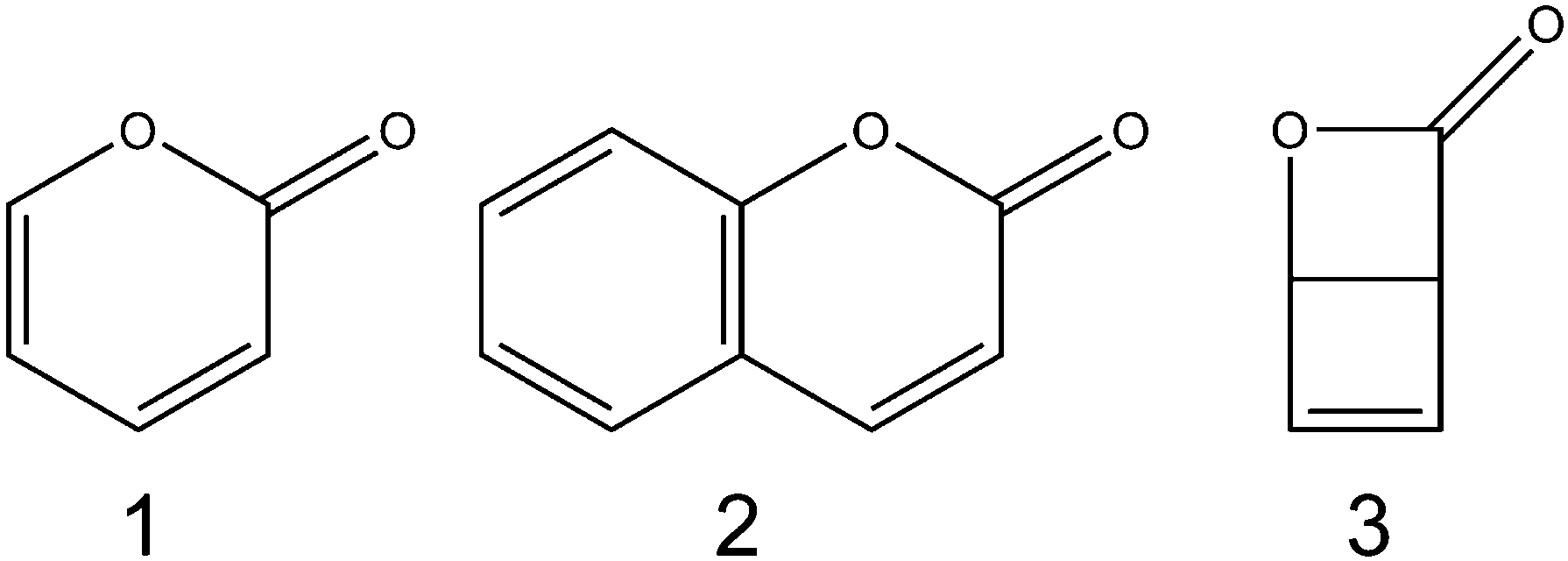
Contrasting ring-opening propensities in UV-excited α-pyrone and coumarin - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/C5CP06597F

α-pyrones: Small molecules with versatile structural diversity reflected in multiple pharmacological activities-an update - ScienceDirect
![The [4 + 2] Cycloaddition of 2‐Pyrone in Total Synthesis - Cai - 2019 - Chinese Journal of Chemistry - Wiley Online Library The [4 + 2] Cycloaddition of 2‐Pyrone in Total Synthesis - Cai - 2019 - Chinese Journal of Chemistry - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/527c5a71-d185-4f29-8a6e-a71d8c51c900/cjoc201900048-fig-0005-m.jpg)
The [4 + 2] Cycloaddition of 2‐Pyrone in Total Synthesis - Cai - 2019 - Chinese Journal of Chemistry - Wiley Online Library

Pyrone-derived Marine Natural Products: A Review on Isolation, Bio-activities and Synthesis | Bentham Science
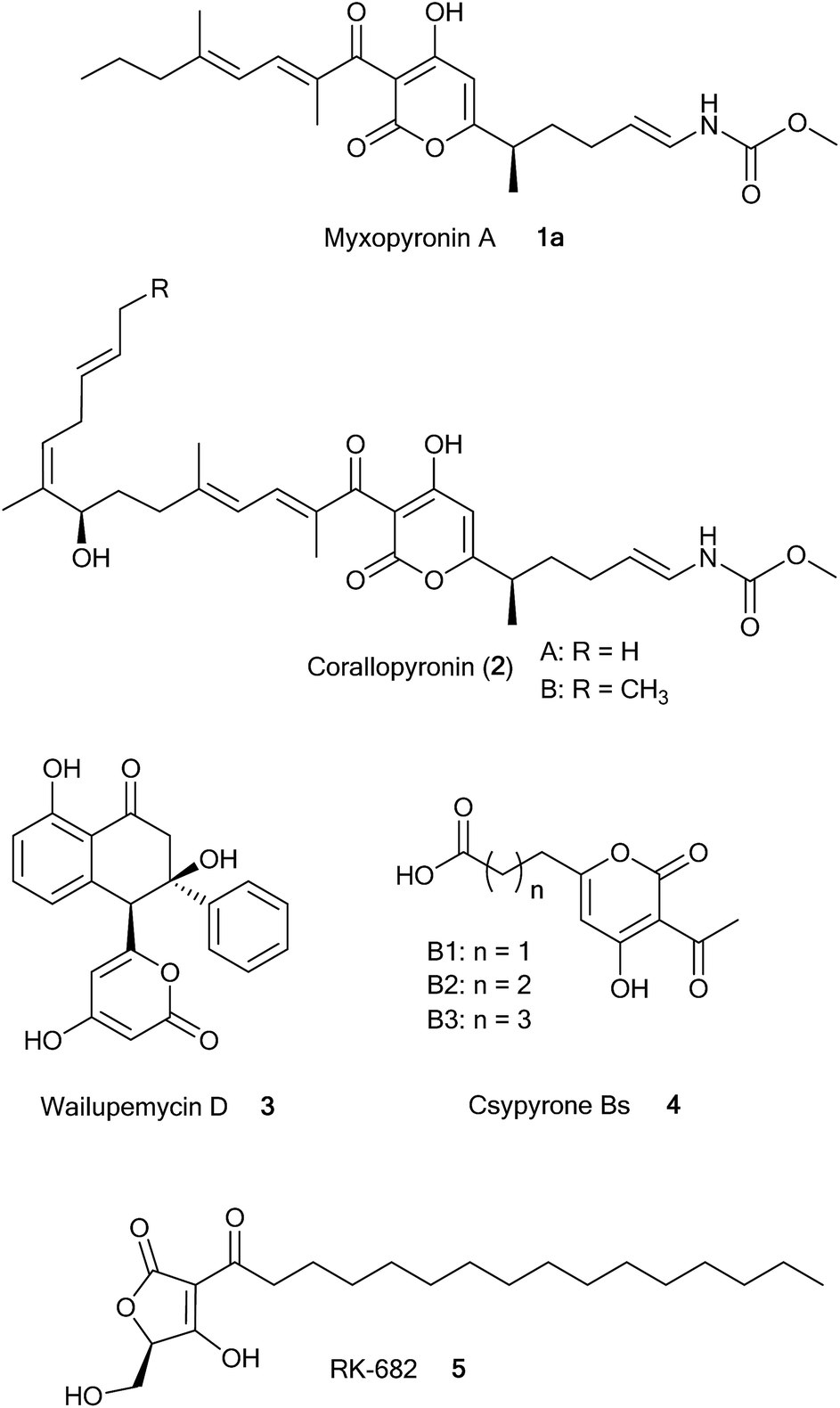
In vitro reconstitution of α-pyrone ring formation in myxopyronin biosynthesis - Chemical Science (RSC Publishing) DOI:10.1039/C5SC01013F