The rate constant for the decompoistion of a certain reaction is described by the equation: log k(s^(-1)) = 14 - (1.25 xx 10^(4) K)/(T) A two-step mechanism has been suggested for the

Chpt 12 - Chemical Kinetics Reaction Rates Rate Laws Reaction Mechanisms Collision Theory Catalysis HW set1: Chpt 12 - pg , # 22, 23, 28 Due Jan. - ppt download
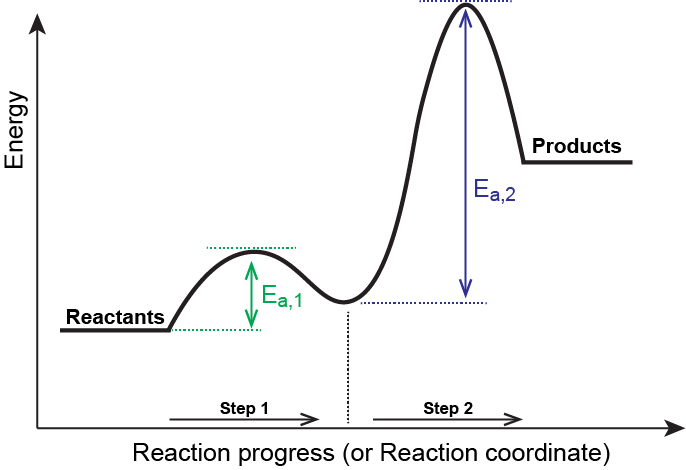
M13Q10: Mechanisms and Multistep Reactions; Reaction Profiles; Rate Limiting Steps – Chem 103/104 Resource Book