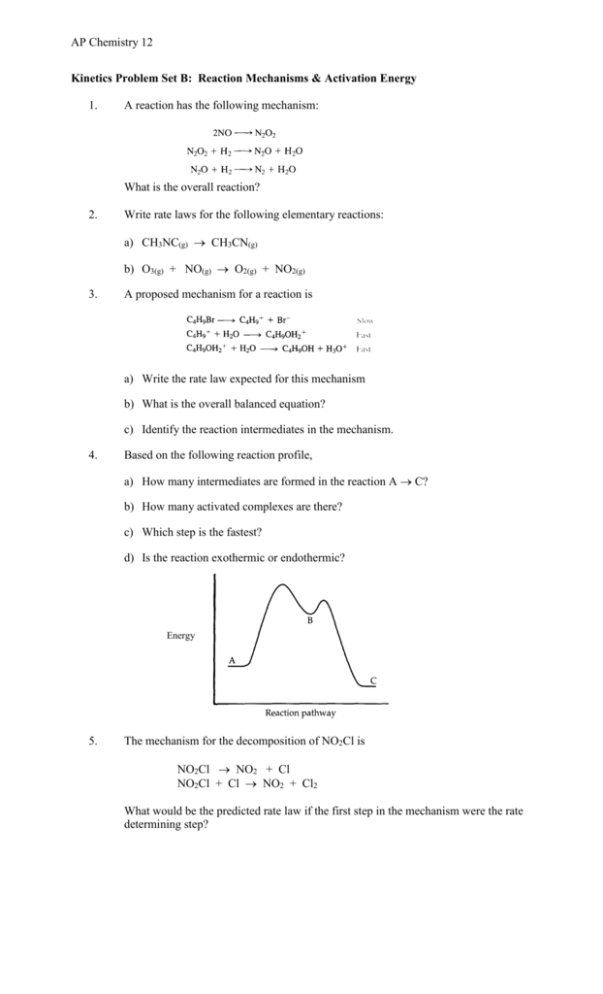
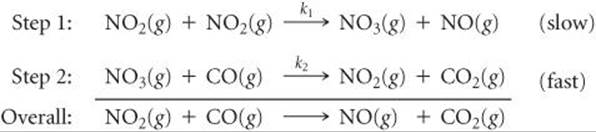
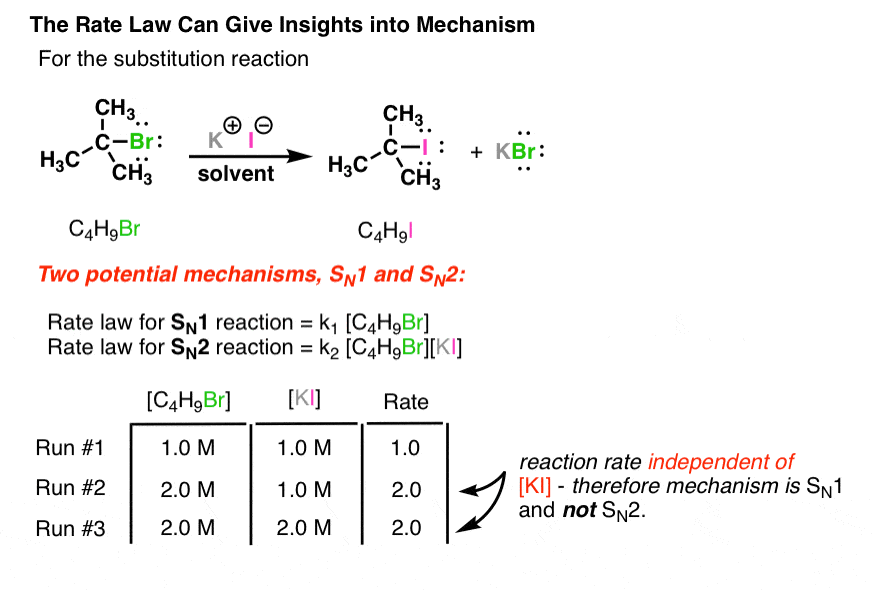
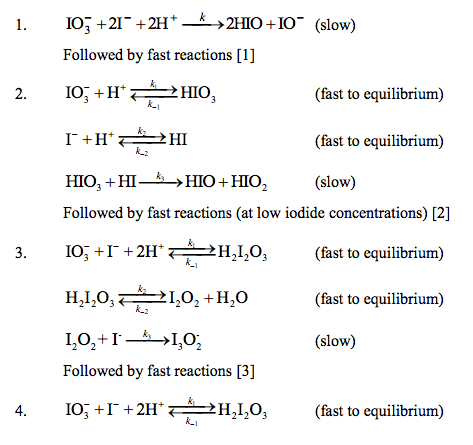Determine the rate law for the overall reaction (where the overall rate constant is represented as k)? | Socratic
![Consider the following mechani[{Image src='img137723731702631852348.jpg' alt='' caption=''}]sm. Determine the rate law for the overall reaction (where the overall rate constant is represented as k). | Study.com Consider the following mechani[{Image src='img137723731702631852348.jpg' alt='' caption=''}]sm. Determine the rate law for the overall reaction (where the overall rate constant is represented as k). | Study.com](https://study.com/cimages/multimages/16/img137723731702631852348.jpg)
Consider the following mechani[{Image src='img137723731702631852348.jpg' alt='' caption=''}]sm. Determine the rate law for the overall reaction (where the overall rate constant is represented as k). | Study.com

Finding the rate law from a rate-determining step that contains species not in the overall reaction : r/chemhelp

SOLVED:The reaction mechanism Step A2 A + A assume fast reversible A + B P slow The slow step involves an intermediate A_ Deduce a rate law for the formation of P_

Rate Determining Step - Rate Processes In Chemical Reactions Kinetics And Equilibrium - MCAT Content

Top of Page Periodic Table Andover's Chem 550/580: Advanced Chemistry Table of Contents Chapter 19: Chemical Kinetics Section 19-1: Collision Theory and Factors That Increase Chemical Reaction Rates Section 19-2: Reaction Energy Profiles ...

SOLVED:The accompanying image shows a possible reaction mechanism for the formation of oxygen from ozone: Choose the rate law predicted by this mechanism. [Image description: First step (slow): O3 7 Oz +