Efficient preparation of dichloromethyl alkyl ethers and their application in the formylation of aromatic compounds: Scope and limitations - ScienceDirect

Palladium-catalyzed C-H formylation of electron-rich heteroarenes through radical dichloromethylation - ScienceDirect

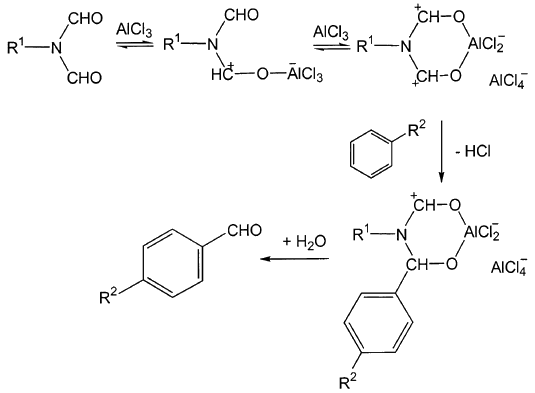
Oxidation‐Induced para‐Selective Formylation of N,N‐Substituted Aniline - Sun - 2018 - Asian Journal of Organic Chemistry - Wiley Online Library
![Redox neutral [4+2] benzannulation of dienals and tertiary enaminones for benzaldehyde synthesis - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC03514H Redox neutral [4+2] benzannulation of dienals and tertiary enaminones for benzaldehyde synthesis - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC03514H](https://pubs.rsc.org/image/article/2018/CC/c8cc03514h/c8cc03514h-f1_hi-res.gif)
Redox neutral [4+2] benzannulation of dienals and tertiary enaminones for benzaldehyde synthesis - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC03514H
Sciencemadness Discussion Board - Aromatic aldehydes using Gattermann Koch Reaction - Powered by XMB 1.9.11

Metal-free selective aryl C–H formylation co-controlled by 1,2,3-triazole and hydroxyl using DMSO as formyl source | SpringerLink

Summary of the molecular structures and field-effect mobility measured... | Download Scientific Diagram
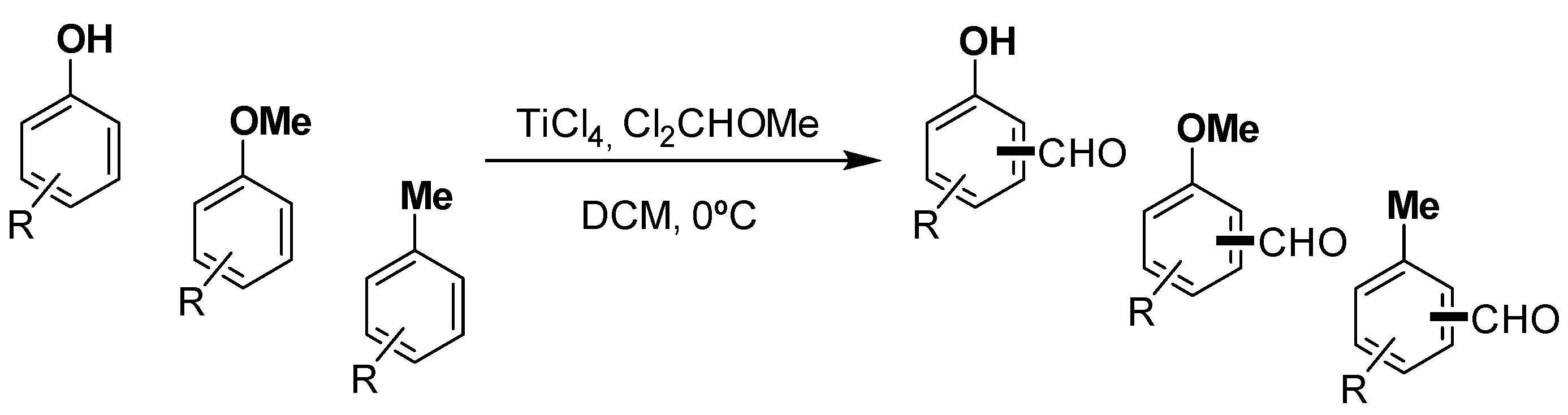
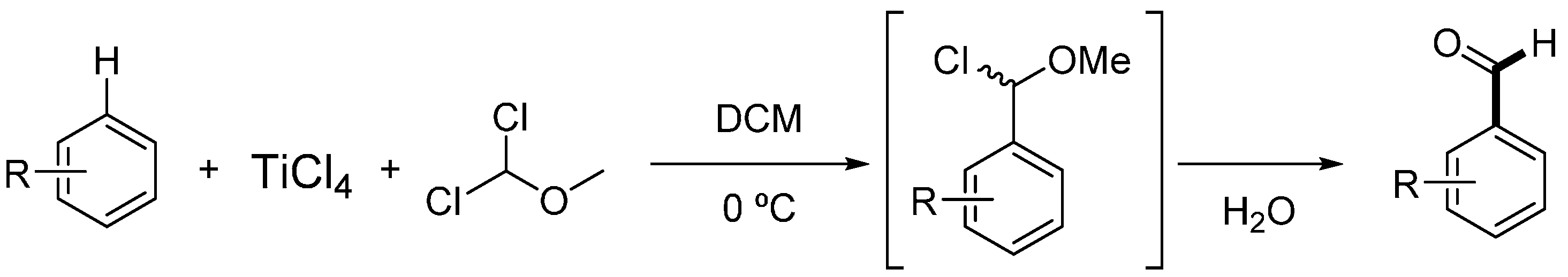
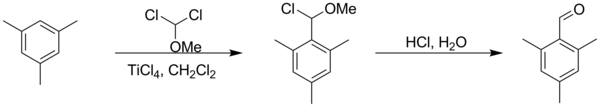
Molecules | Free Full-Text | Formylation of Electron-Rich Aromatic Rings Mediated by Dichloromethyl Methyl Ether and TiCl4: Scope and Limitations | HTML

Efficient preparation of dichloromethyl alkyl ethers and their application in the formylation of aromatic compounds: Scope and limitations - ScienceDirect

Gold(I)-Mediated Cycloisomerization/Cycloaddition Enables Bioinspired Syntheses of Neonectrolides B-E and Analogues. - Abstract - Europe PMC

Efficient preparation of dichloromethyl alkyl ethers and their application in the formylation of aromatic compounds: Scope and limitations - ScienceDirect

Polyketide Cyclizations for the Synthesis of Polyaromatics - Fäseke - 2020 - Angewandte Chemie - Wiley Online Library
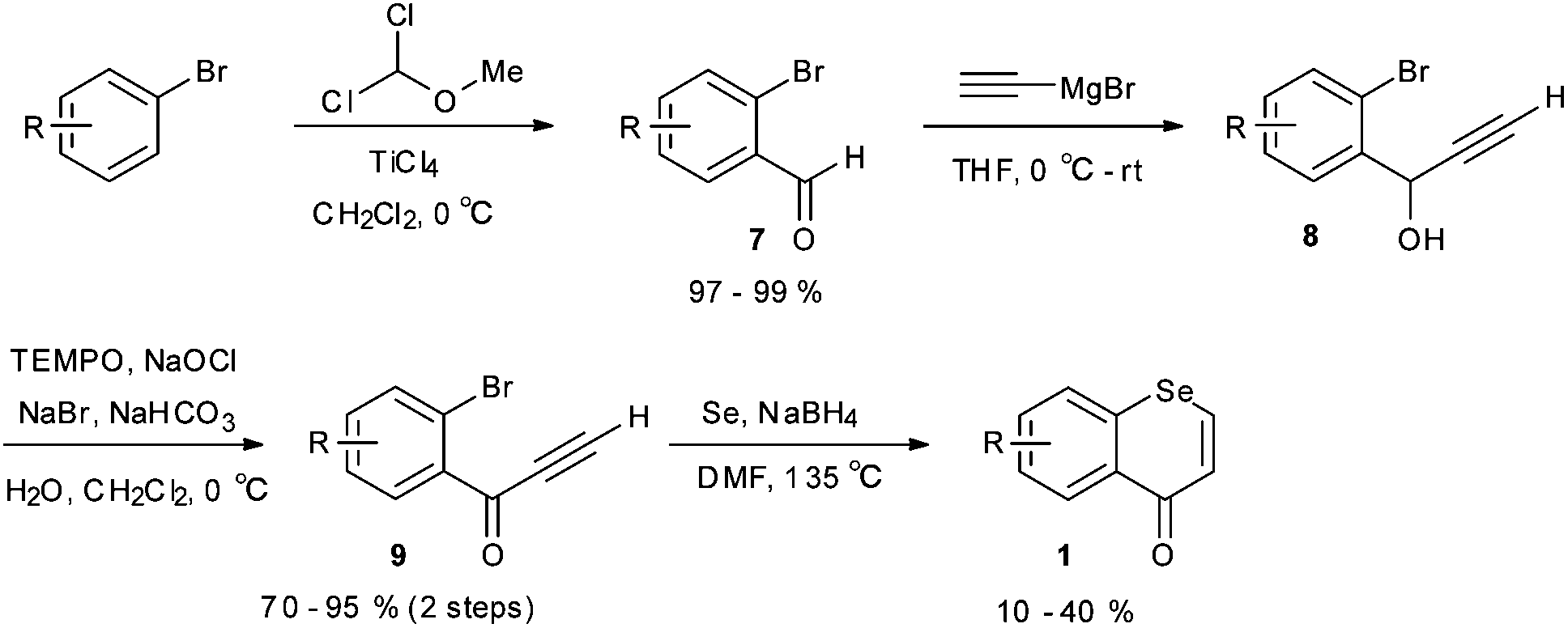
Efficient synthesis of polymethoxyselenoflavones via regioselective direct C–H arylation of selenochromones - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB00118E