
Structure of the C-terminal RING Finger from a RING-IBR-RING/TRIAD Motif Reveals a Novel Zinc-binding Domain Distinct from a RING - ScienceDirect

Characterisation of class VI TRIM RING domains: linking RING activity to C-terminal domain identity | Life Science Alliance

The RING finger- and PDZ domain-containing protein PDZRN3 controls localization of the Mg2+ regulator claudin-16 in renal tube epithelial cells - Journal of Biological Chemistry

Membranes | Free Full-Text | The Role of the Transmembrane RING Finger Proteins in Cellular and Organelle Function | HTML
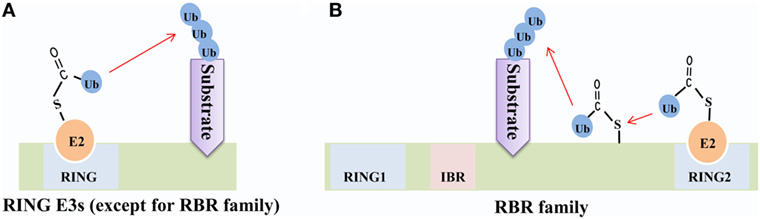
Frontiers | RING-Domain E3 Ligase-Mediated Host–Virus Interactions: Orchestrating Immune Responses by the Host and Antagonizing Immune Defense by Viruses | Immunology
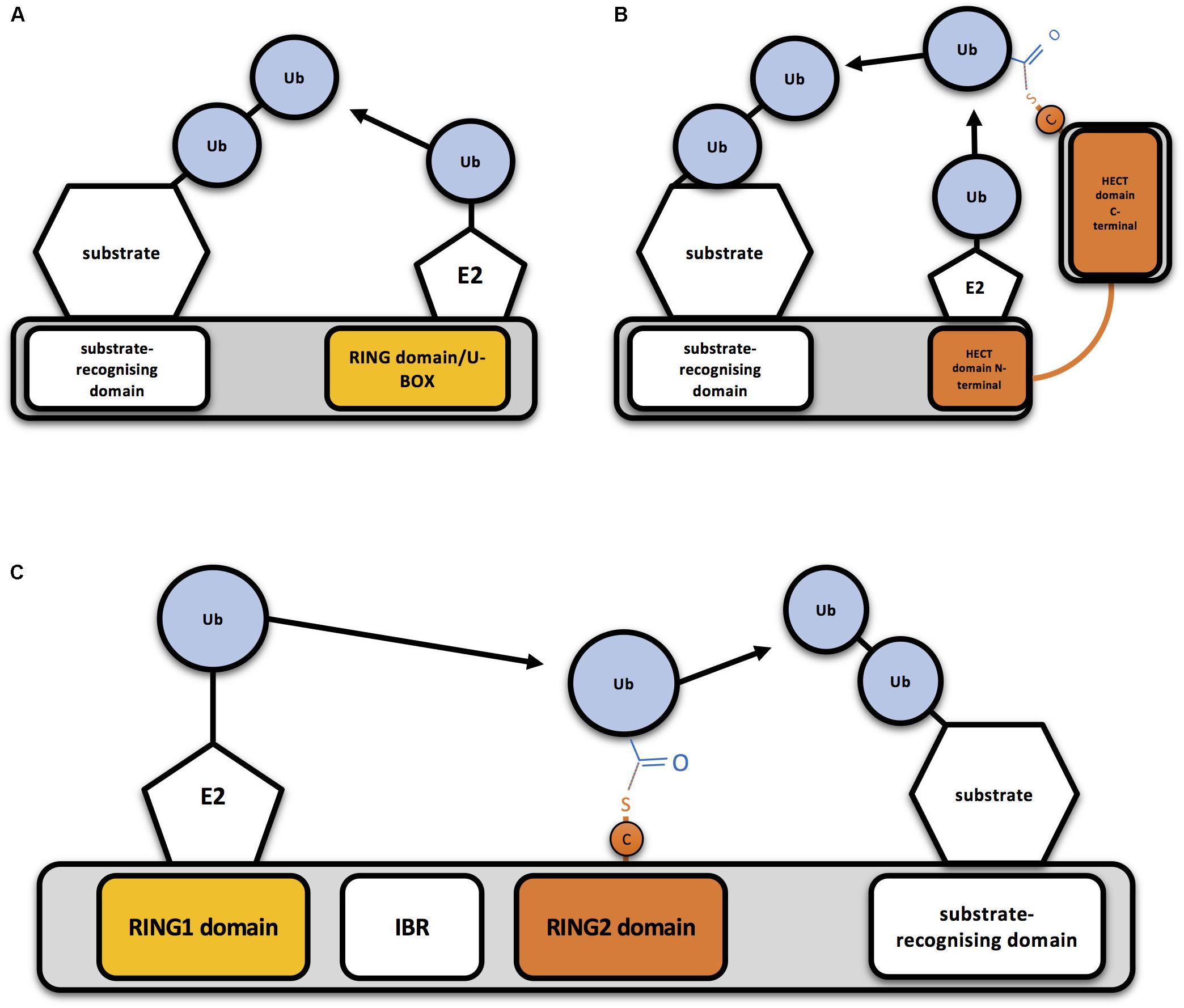
Frontiers | How to Inactivate Human Ubiquitin E3 Ligases by Mutation | Cell and Developmental Biology

Solution structure of MUL1-RING domain and its interaction with p53 transactivation domain - ScienceDirect

A central role for the ring finger protein RNF11 in ubiquitin-mediated proteolysis via interactions with E2s and E3s | Oncogene
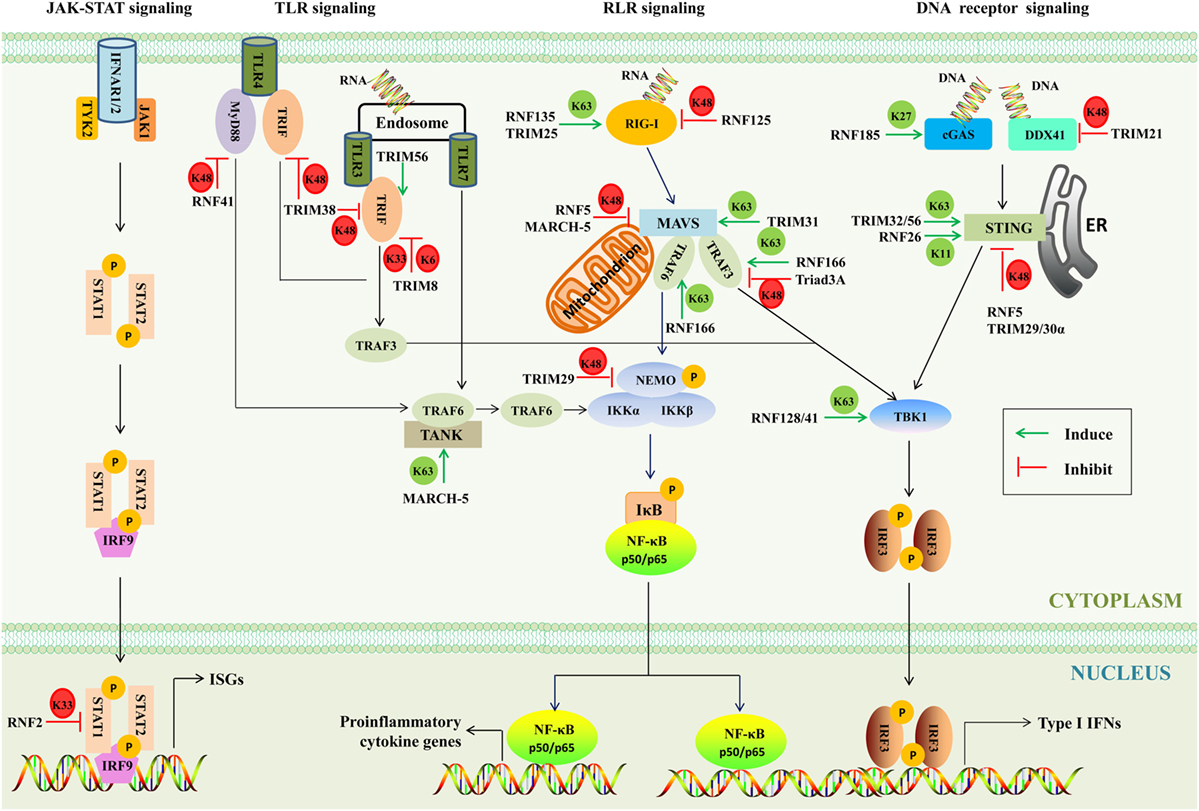
Frontiers | RING-Domain E3 Ligase-Mediated Host–Virus Interactions: Orchestrating Immune Responses by the Host and Antagonizing Immune Defense by Viruses | Immunology
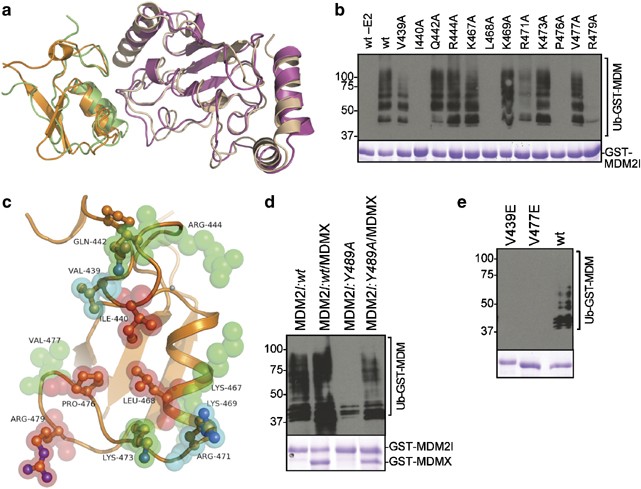
Structure of the MDM2/MDMX RING domain heterodimer reveals dimerization is required for their ubiquitylation in trans | Cell Death & Differentiation
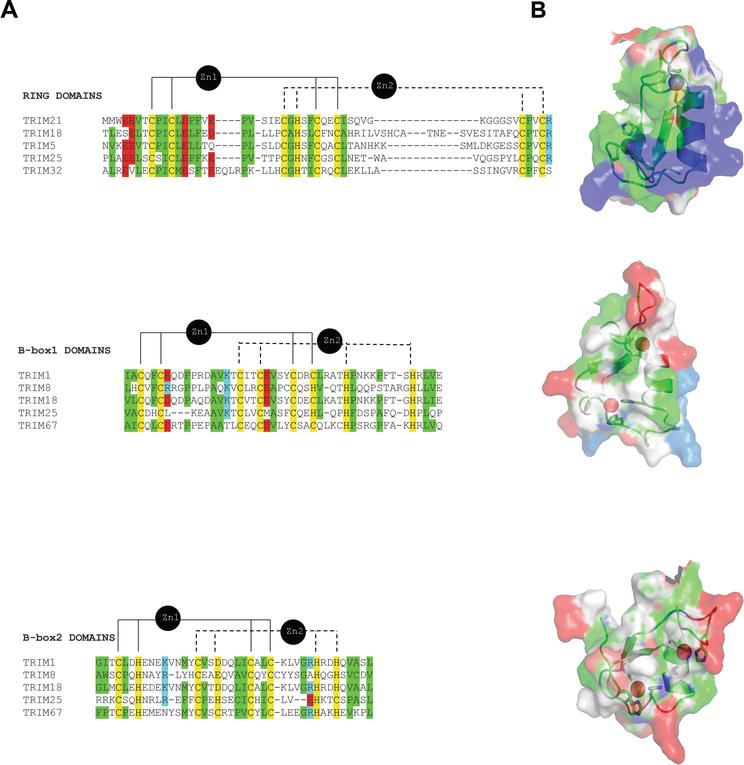
Zinc-Binding B-Box Domains with RING Folds Serve Critical Roles in the Protein Ubiquitination Pathways in Plants and Animals | IntechOpen
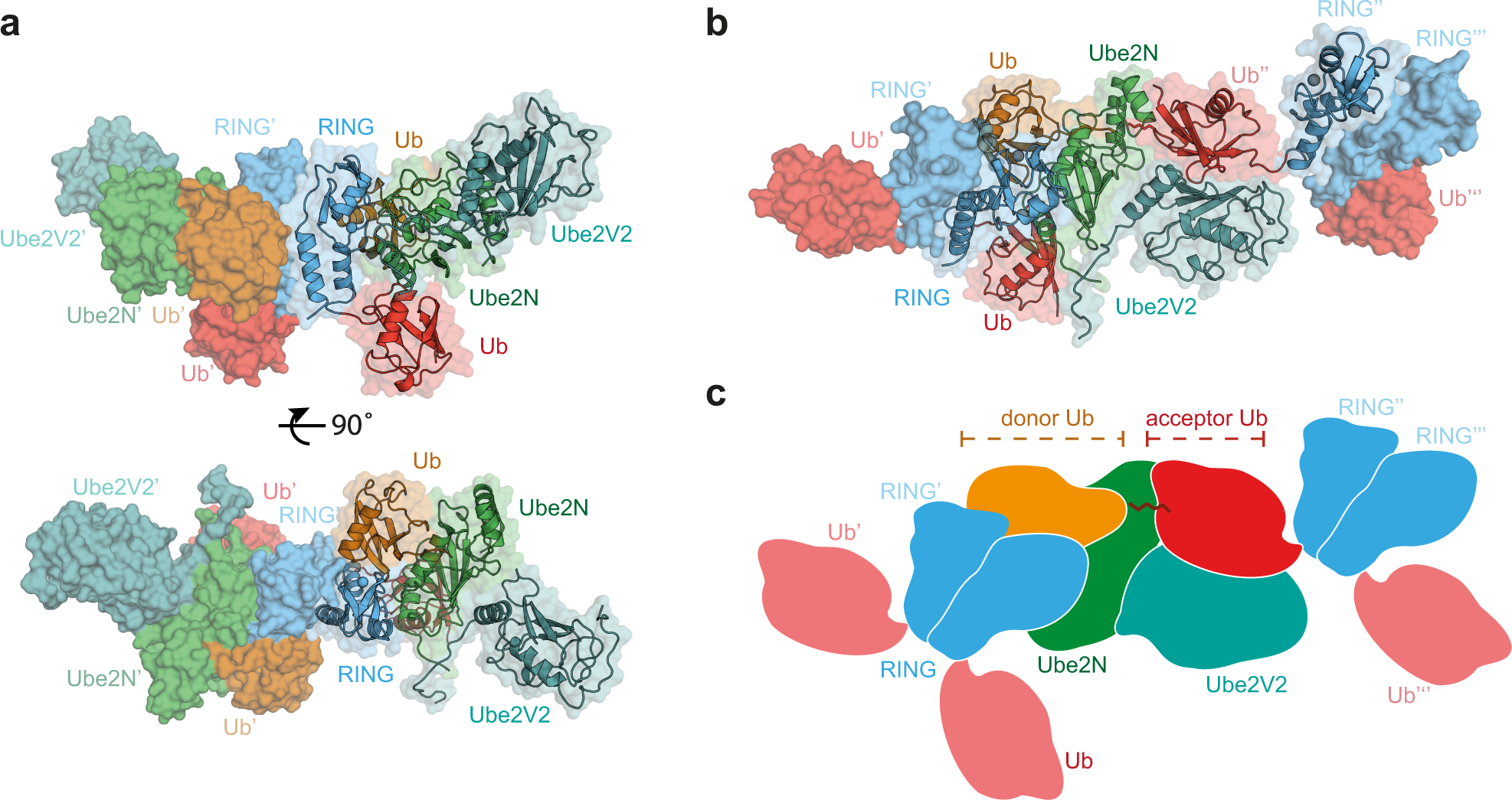




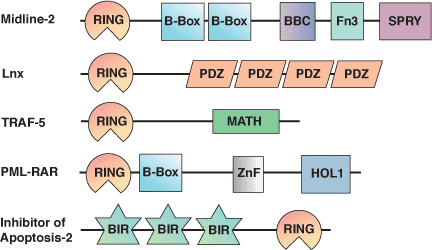

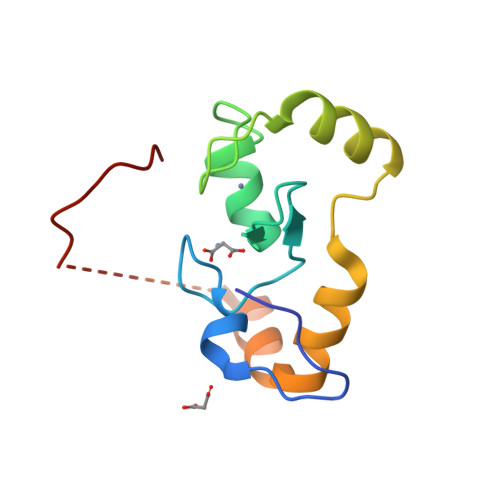



![PDF] RING domain E3 ubiquitin ligases. | Semantic Scholar PDF] RING domain E3 ubiquitin ligases. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bad3f08c363426e09b0089582deb2cecfa364e86/10-Figure5-1.png)


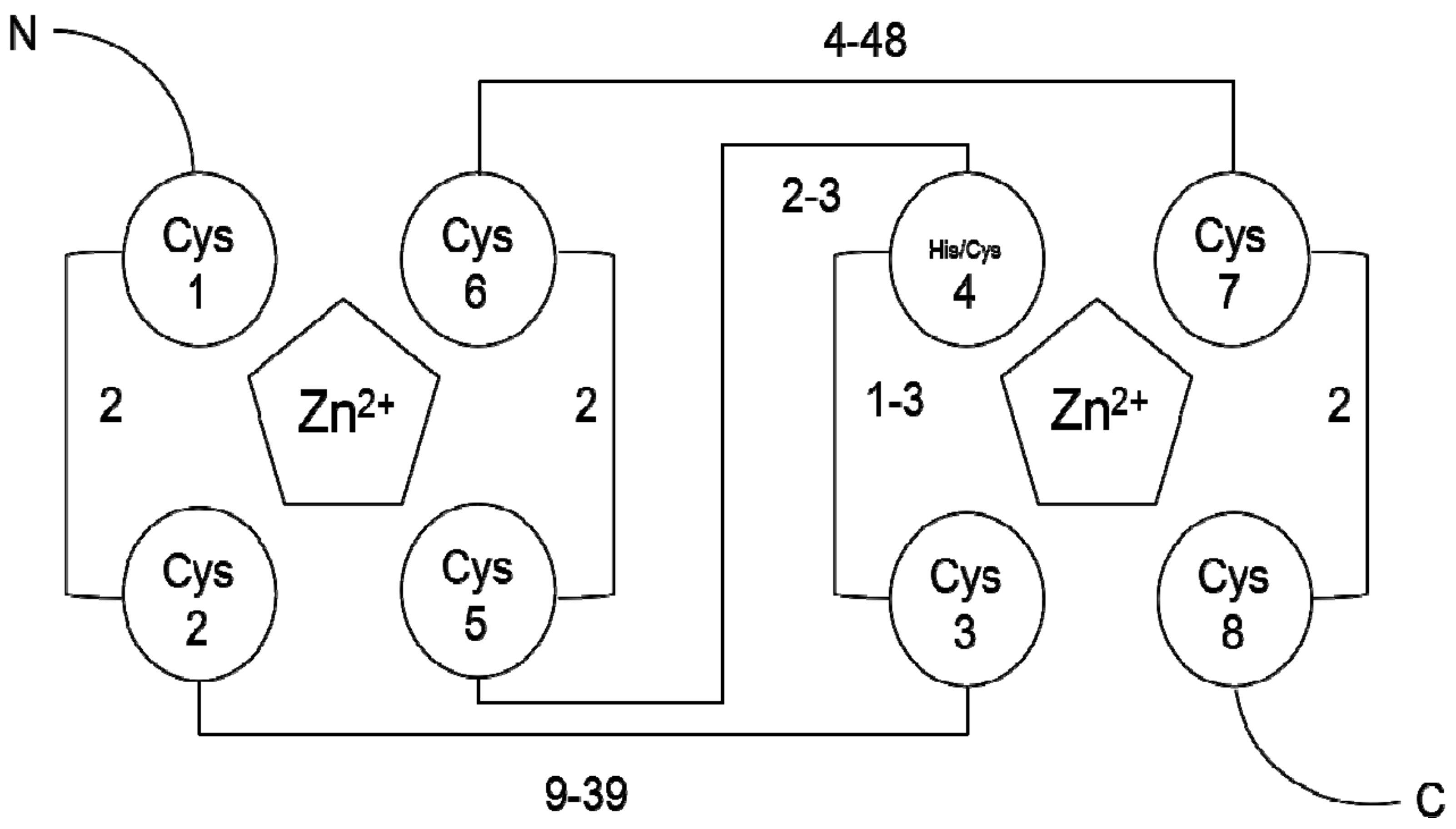
![PDF] RING domain E3 ubiquitin ligases. | Semantic Scholar PDF] RING domain E3 ubiquitin ligases. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/bad3f08c363426e09b0089582deb2cecfa364e86/3-Figure1-1.png)
