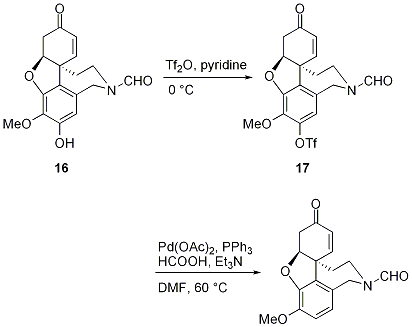
First Catalytic and Green Synthesis of Aryl-(Z)-vinyl Chlorides and Its Plausible Addition-Elimination Mechanism
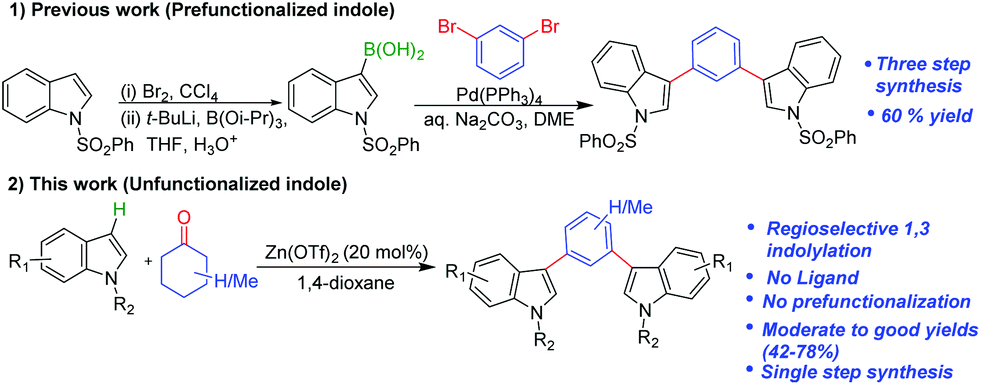
Zinc triflate catalyzed 1,3-indolylation of cyclohexanones: tandem condensation, dehydrogenation and aromatization sequence - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/D0OB00163E

Metal triflates are tunable acidic catalysts for high yielding conversion of cellulosic biomass into ethyl levulinate - ScienceDirect

Scheme 2. Synthesis of two triflate precursors 6 and 10. Conditions:... | Download Scientific Diagram
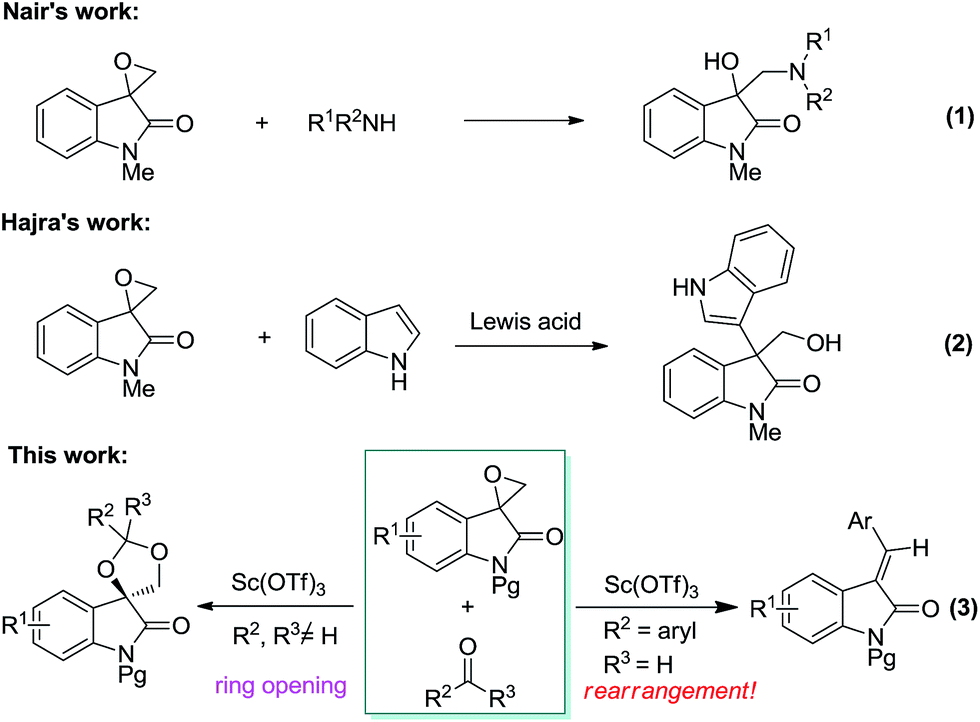
Scandium triflate-catalyzed selective ring opening and rearrangement reaction of spiro-epoxyoxindole and carbonyl compounds - RSC Advances (RSC Publishing) DOI:10.1039/C6RA21264F

Mechanistic studies on a sulfoxide transfer reaction mediated by diphenyl sulfoxide/triflic anhydride. - Abstract - Europe PMC
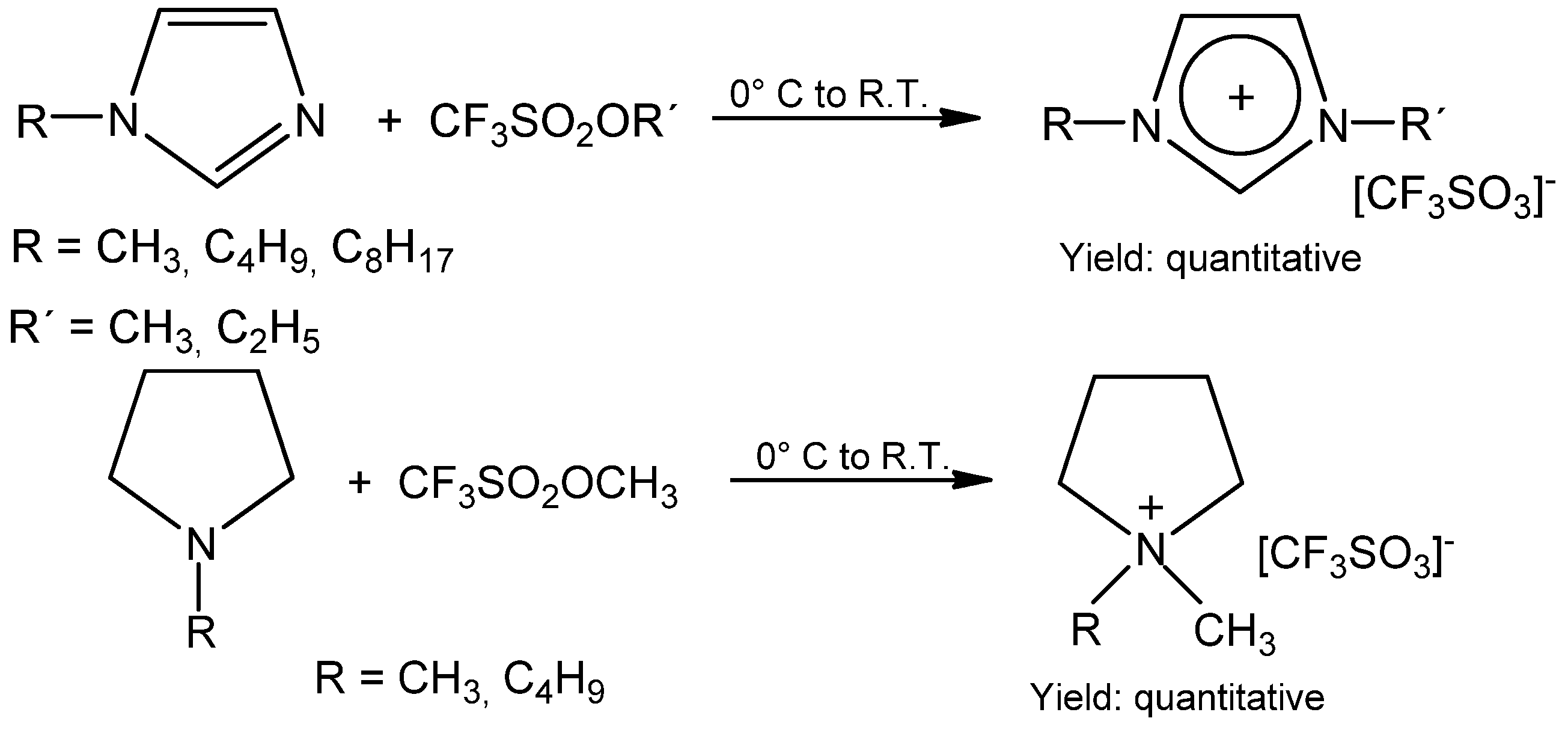
Molecules | Free Full-Text | A Convenient Synthesis of Triflate Anion Ionic Liquids and Their Properties | HTML
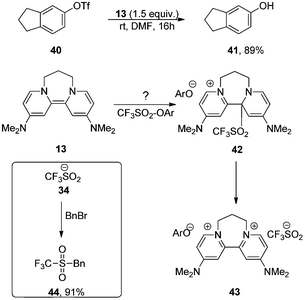
Reactions of triflate esters and triflamides with an organic neutral super-electron-donor - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB25116G
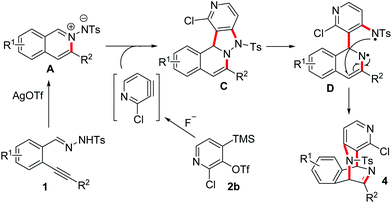
Silver triflate-catalyzed tandem reaction of N ′-(2-alkynylbenzylidene)hydrazide with pyridyne - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C2OB26379C

Proposed mechanism demonstrating synergistic reactions between triflate... | Download Scientific Diagram
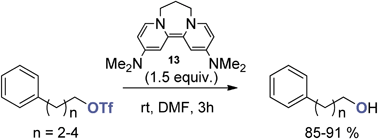
Reactions of triflate esters and triflamides with an organic neutral super-electron-donor - Organic & Biomolecular Chemistry (RSC Publishing)
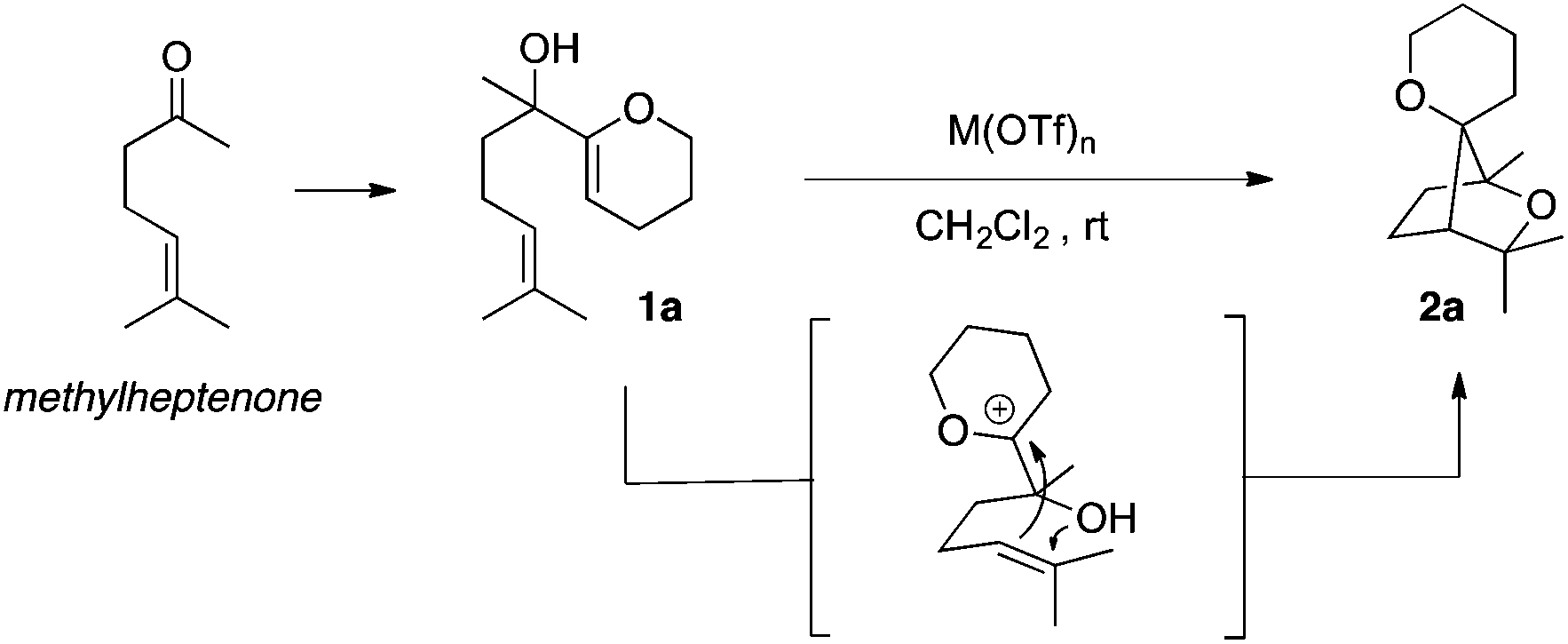
Bismuth( iii ) triflate-catalysed tandem cyclisations towards complex polycyclic ethers - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C6QO00212A
![Hafnium Trifluoromethanesulfonate [Hf(OTf)4] as a Unique Lewis Acid in Organic Synthesis - Ishitani - 2015 - European Journal of Organic Chemistry - Wiley Online Library Hafnium Trifluoromethanesulfonate [Hf(OTf)4] as a Unique Lewis Acid in Organic Synthesis - Ishitani - 2015 - European Journal of Organic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/c31f8538-fb8f-4037-8ce4-24694af59f3d/mfig000.jpg)
Hafnium Trifluoromethanesulfonate [Hf(OTf)4] as a Unique Lewis Acid in Organic Synthesis - Ishitani - 2015 - European Journal of Organic Chemistry - Wiley Online Library