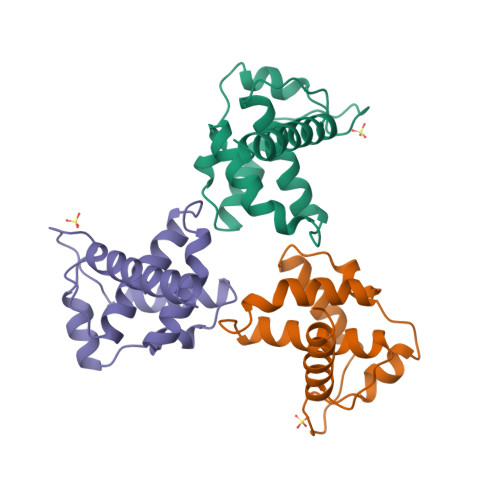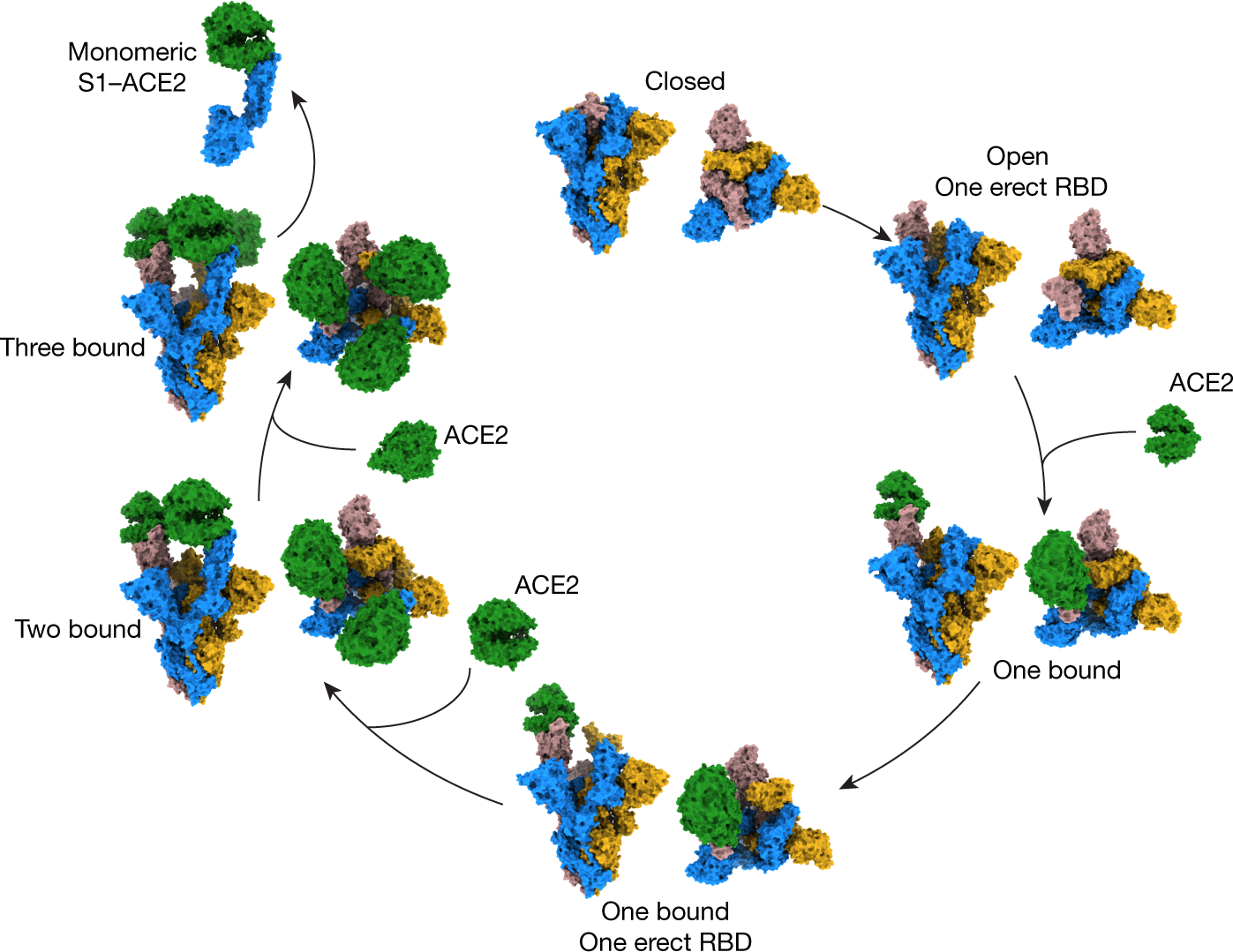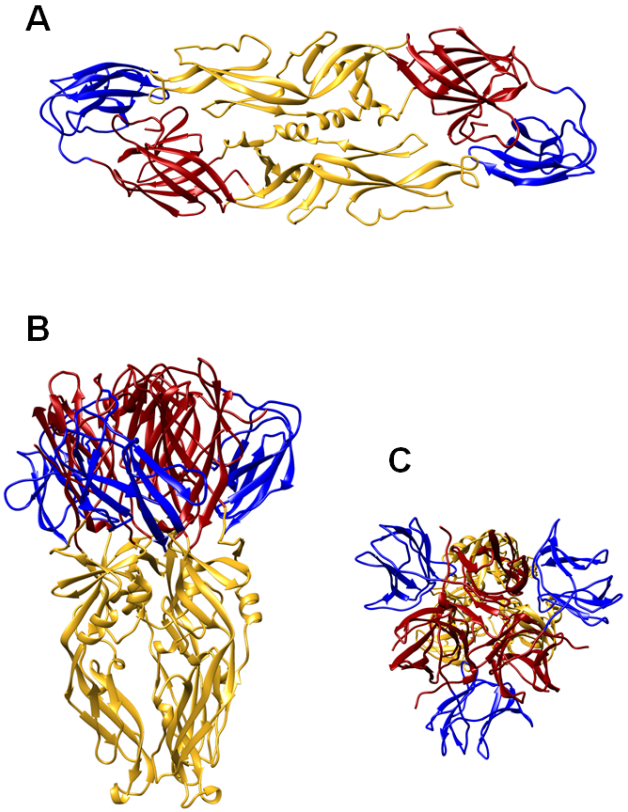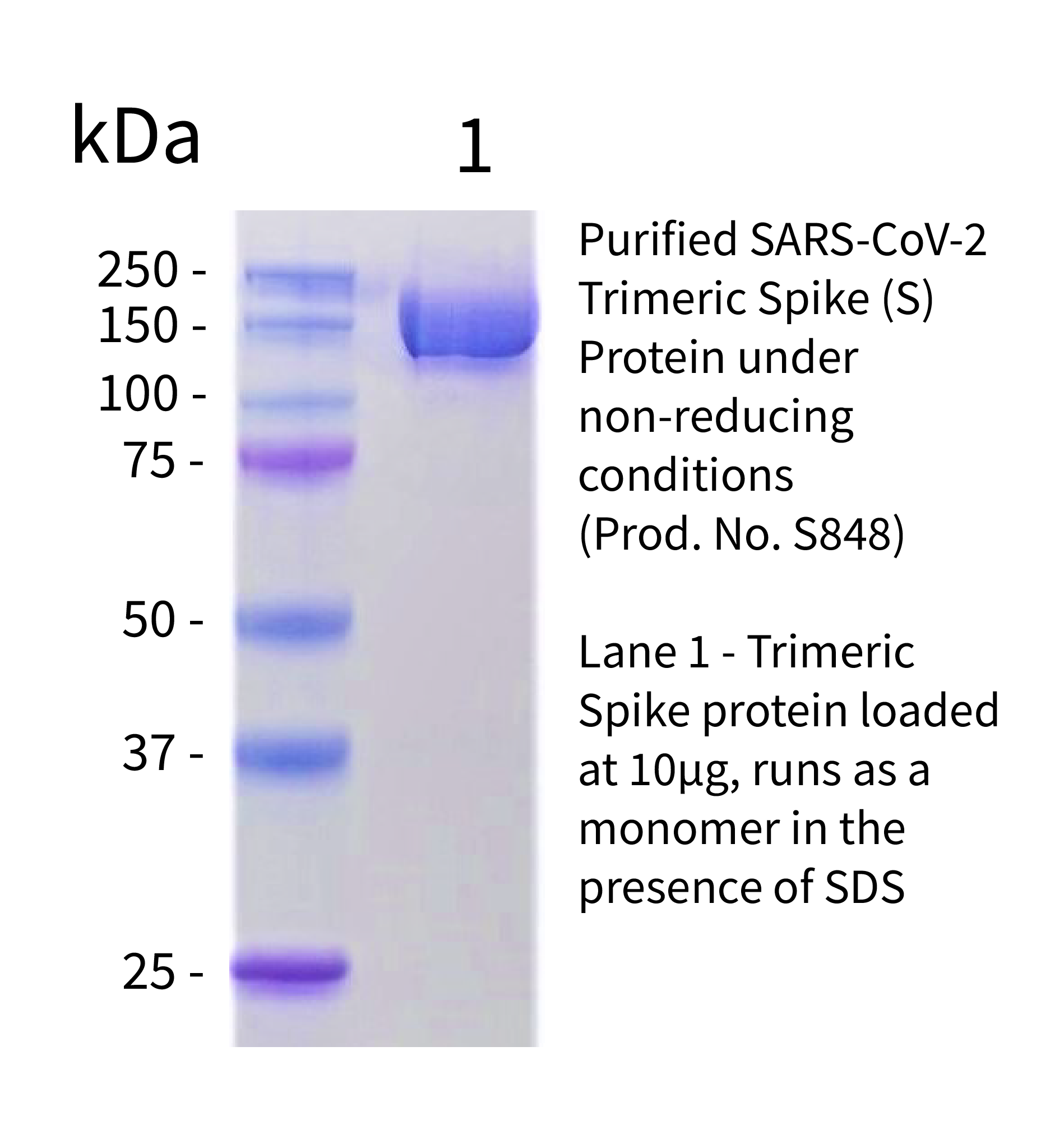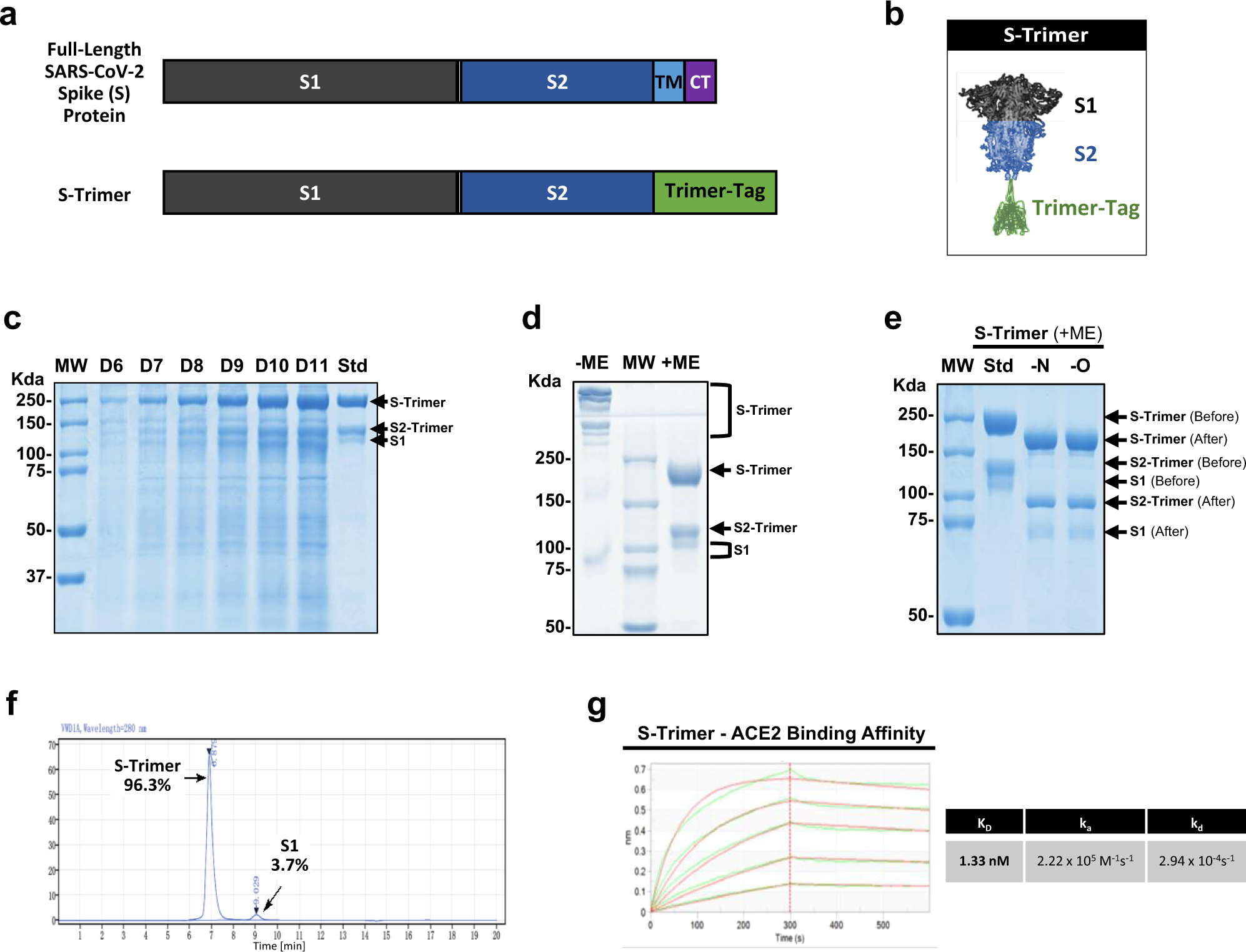
S-Trimer, a COVID-19 subunit vaccine candidate, induces protective immunity in nonhuman primates | Nature Communications

Actinobacteria challenge the paradigm: A unique protein architecture for a well-known, central metabolic complex | PNAS
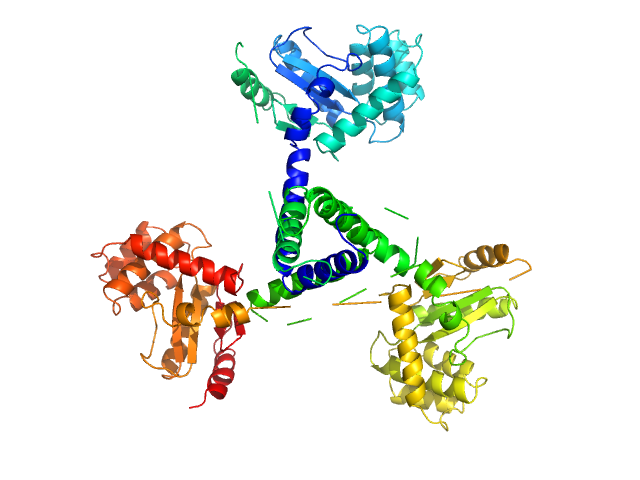
SASDER4 – Wild-type suppressor of copper sensitivity C protein, PmScsC, with concentration series data

Metal‐Templated Design of Chemically Switchable Protein Assemblies with High‐Affinity Coordination Sites - Kakkis - 2020 - Angewandte Chemie - Wiley Online Library

Comparison of the functional properties of trimeric and monomeric CaiT of Escherichia coli | Scientific Reports

Trimeric Structure of (+)-Pinoresinol-forming Dirigent Protein at 1.95 Å Resolution with Three Isolated Active Sites* - Journal of Biological Chemistry

Crystal Structure of Trimeric Carbohydrate Recognition and Neck Domains of Surfactant Protein A* - Journal of Biological Chemistry

Production and stabilization of the trimeric influenza hemagglutinin stem domain for potentially broadly protective influenza vaccines | PNAS
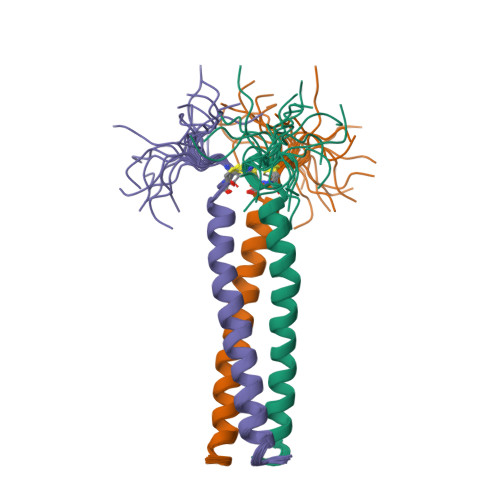
RCSB PDB - 1AQ5: HIGH-RESOLUTION SOLUTION NMR STRUCTURE OF THE TRIMERIC COILED-COIL DOMAIN OF CHICKEN CARTILAGE MATRIX PROTEIN, 20 STRUCTURES
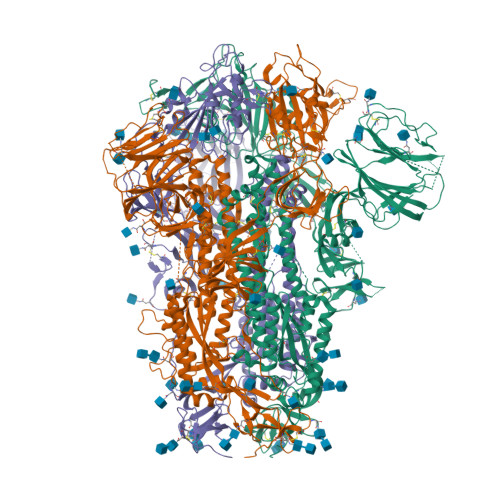
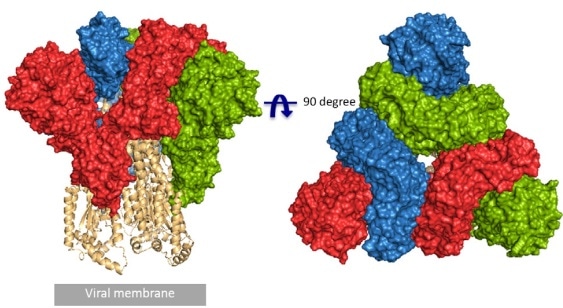
RCSB PDB - 6ZP1: Structure of SARS-CoV-2 Spike Protein Trimer (K986P, V987P, single Arg S1/S2 cleavage site) in Closed State

Revealing the Activity of Trimeric G-proteins in Live Cells with a Versatile Biosensor Design - ScienceDirect
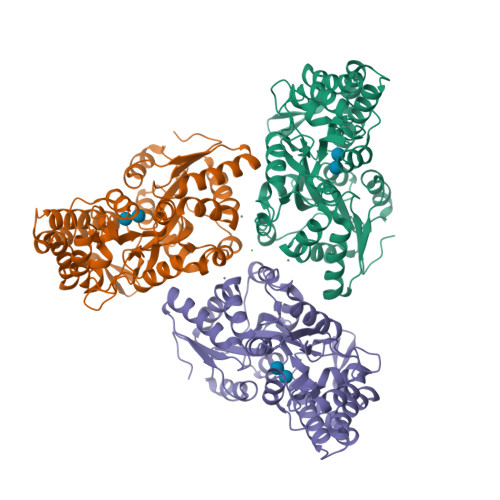
RCSB PDB - 3SEV: Zn-mediated Trimer of Maltose-binding Protein E310H/K314H by Synthetic Symmetrization

Active Participation of Hsp90 in the Biogenesis of the Trimeric Reovirus Cell Attachment Protein ς1* - Journal of Biological Chemistry